Overview
Diabetes Canada Clinical Practice Guidelines Expert Committee
James A. Stone MD, PhD, FRCPC, Robyn L. Houlden MD, FRCPC, Peter Lin MD, CCFP, Jacob A. Udell MD, MPH, FRCPC, Subodh Verma MD, PhD, FRCSC, FAHA
Anchored List of chapter sections
1. Key Messages
- Diabetes significantly accelerates the development and natural history of cardiovascular disease compared to individuals without diabetes.
- Healthy behaviour interventions and pharmacological approaches aimed at cardiovascular disease risk reduction can significantly reduce morbidity and mortality, and are an important cornerstone of the management of diabetes.
- Although young people with diabetes rarely have a high proximate (<10 year) risk for cardiovascular disease events, they have a relative proximate risk manyfold greater than individuals of similar age without diabetes.
- Historically, pharmacological cardiovascular protection approaches have focused on low-density lipoprotein cholesterol and blood pressure reduction, and have demonstrated significant and clinically meaningful cardiovascular risk reduction. Recent data have indicated that certain antihyperglycemic agents are also cardioprotective.
- The requirement for pharmacological cardiovascular protection therapies (statins, angiotensin-converting enzyme inhibitors or aldosterone receptor blockers, and anti-platelets) should consider both an individual's proximate and lifetime cardiovascular disease event risk.
- There is emerging recognition that nonatherothrombotic cardiovascular disease complications, such as heart failure, are an important cause of morbidity and mortality in diabetes.
2. Key Messages for People with Diabetes
- Ask your doctor about the ABCDEs to reduce your risk of heart attack and stroke:
- A = A1C – Blood glucose control. The target is usually 7.0% or less.
- B = BP – Blood pressure control (less than 130/80 mmHg).
- C = Cholesterol – LDL-cholesterol less than 2.0 mmol/L. Your physician/nurse practitioner may advise you to start cholesterol-lowering medication.
- D = Drugs to protect your heart – These include blood pressure pills (ACE inhibitors or ARBs), cholesterol-lowering medication (“statins”), and, in people with existing cardiovascular disease, certain blood glucose lowering medications. These blood glucose-lowering medications can protect your heart even if your blood pressure and/or LDL-cholesterol are already at target.
- E = Exercise/Eating — Regular physical activity, which includes healthy eating, and achievement and maintenance of a healthy body weight.
- S = Stop smoking and manage stress.
3. Introduction
Of the many complex complications of diabetes, adverse cardiovascular (CV) events have the greatest capacity to cause sudden or premature death and devastating disability. Myocardial infarction (MI), stroke and amputation are all manifestations of the aggressive atherosclerosis that can occur with diabetes. However, not every person with diabetes is at equal risk for CV atherosclerotic events and not everyone will benefit equally from healthy behaviour and pharmacological interventions intended to reduce cardiovascular disease (CVD) event risk. Over the last 2 decades, strong evidence has continued to accumulate that the CV risks of diabetes can be reduced significantly through comprehensive and treatment target-driven risk factor modification (1–5). There is also growing appreciation that, in addition to atherothrombotic consequences, other CV disorders, such as heart failure, are an important cause of morbidity and mortality in diabetes.
4. Determining CVD Event Risk
People with diabetes are clearly at increased risk of premature morbidity and mortality related to CVD (6). Diabetes confers a CVD event risk that is equivalent to aging approximately 15 years, with a transition from intermediate to high risk in men at age 47.9 years, and in women at 54.3 years (6). The term “vascular age” refers to models of CVD event risk that predict an individual's CVD event risk and compare the event risk to age-adjusted CVD event risk. Vascular age is a primary determinant in both proximate (<10 years) and lifetime risk of adverse CVD events. In people with diabetes with low-to-normal levels of blood pressure (BP), low-density lipoprotein cholesterol (LDL-C) and blood glucose (BG), chronological age and vascular age are usually in close continuity. However, in the presence of elevated levels of those same variables, together with smoking and physical inactivity, vascular age accelerates far more rapidly than chronological age.
As a powerful catalyst of vascular inflammation, diabetes is the disease state that accelerates vascular age at the greatest rate. Thus, the use of pharmacotherapy for CVD risk factor reduction in younger persons with diabetes who are not at a high proximate risk but, as a consequence of their diabetes, have a steep CVD event risk trajectory, can be justified by the potentially substantial long-term benefits of earlier and lifelong therapy (3–5,7,8).
Traditional CVD event risk models predict an individual's proximate CVD event risk based on risk factors, such as diabetes, hypertension, serum lipids and smoking. These models discriminate poorly between higher- and lower-risk populations, particularly for younger individuals (9–12). In addition, no current CVD event risk model can reliably exclude people with diabetes who are unlikely to benefit from long-term CV protection strategies given the well-documented lifetime risk of CVD events. As a result, far in advance of the appearance of CV symptomatology, most people with diabetes are very likely to benefit from CVD risk factor reduction and the adoption of healthy behaviours (3–5,7,8).
5. Cardiovascular Protection
The phrase “vascular protection” was originally coined in recognition of the apparent ability of some pharmacologic interventions to evoke greater reductions in the incidence of CVD events than would have been predicted based on their separate direct effects on the risk factors for atherosclerosis (13). This putative protective effect has been attributed to the enhancement of vascular endothelial functions that inhibit thrombosis, suppress macrophage and monocyte adherence to the endothelium, and minimize oxidative stress at the level of the endothelium (14). Over time, the protective effect from adverse CVD events has also been extended to comprehensive healthy behaviour interventions and simultaneous multifactorial atherosclerosis risk factor reductions, such that the whole in CV protection is indeed greater than the sum of the parts (3–5). The mechanistic explanation for why multifaceted CV protection interventions are multiplicative, rather than simply additive, is almost certainly related to, but not necessarily limited to, their favourable modulation of the pro-inflammatory, pro-thrombotic and pro-proliferative atherosclerotic vascular environment in diabetes.
In the STENO-2 Trial, a very small number of participants with type 2 diabetes (n=160) were randomized to usual care or a program of comprehensive healthy behaviour interventions (smoking cessation, weight management, physical activity) and the treatment target-driven pharmacological therapy of BP and serum lipids (3,4). Despite the very small number of participants, there was a 53% relative risk reduction in major adverse cardiac events (MACE) and a 20% absolute risk reduction after 13 years of follow up. The number needed to treat (NNT) for mortality reduction was a mere 5 persons. More recently, data from the Bypass Angioplasty Revascularization Investigation 2 Diabetes (BARI 2D) study have demonstrated that, following cardiac revascularization, participants who had their CVD risk factors controlled benefited from significantly improved CVD morbidity and mortality over 5 years of follow up (5). Similarly, in a population-based observational study of 867 newly diagnosed individuals with diabetes, the relative risk of MACE was increased over 4-fold during 5 years of follow up for those persons who adopted no healthy behaviour changes vs. those who adopted either 3 or 4 healthy behaviours (15).
Although the number of participants in these trials is relatively small compared to those in randomized trials of pharmacological agents, the data suggest that efficacious CV protection can be achieved through a combination of healthy behaviours and pharmacologic treatment of CVD risk factors to achieve the targets recommended by evidence-informed clinical practice guidelines (16). Therefore, all people with diabetes should receive a comprehensive, multifactorial strategy to reduce CVD event risk.
6. Strategies for CV Protection
In 1 prospective cohort study of 867 people with newly diagnosed diabetes aged 40 to 69 years, CVD risk was inversely associated with the number of healthy behaviour changes adopted in the year after the diagnosis of diabetes (15). The CV protection benefits of each of the healthy behaviour interventions discussed below can be attributed to their significant anti-inflammatory, antithrombotic and anti-proliferative effects (17).
Smoking cessation
In individuals with diabetes, smoking is an independent risk factor for all-cause mortality. It increases the risk of MI 1.4-fold, stroke by 30% (18), and progression to end stage renal disease (ESRD) (19); and is associated with poorer glycemic control. Quitting smoking has been shown to reduce CV risk in people with diabetes (20).
Physical activity
In several randomized trials, exercise has been shown to improve CV risk factors (dyslipidemia, BP and body composition) in people with type 2 diabetes (21). However, no clinical trials have demonstrated a reduction in major CV endpoints or mortality. The Look AHEAD (Action for Health in Diabetes) trial was the largest randomized trial to date evaluating the efficacy of a physical activity and dietary control intervention (targeting a ≥7% weight loss), in older adults with type 2 diabetes (22). In this study, at least 175 min/week of unsupervised exercise was targeted as part of the Intense Lifestyle Intervention (ILI), while the control group (Diabetes Support and Education-DSE group) received usual care. After a median follow up of 9.6 years, the composite primary outcome (death from CV causes, nonfatal MI, nonfatal stroke and hospitalization for angina) occurred in a similar number of participants in the intervention and control groups (22). Possible reasons for this finding include the lower-than-expected rates of CV events in both groups, improved overall CV risk factor treatment with antihypertensive agents and statins, enrollment of a relatively healthy population and gradual weight loss in the control group (difference in weight loss between the 2 groups was 2.5% at the end of the study). Importantly, and perhaps one explanation for why there was no significant effect on CVD outcomes, after the first year of the trial, the intervention group and the control group were virtually performing the same amounts of exercise and physical activity (see Physical Activity and Diabetes chapter, p. S54).
Several prospective cohort studies have shown that physical activity is associated with improvement in CV outcomes and a reduction in CV and overall mortality in people with type 2 diabetes or impaired glucose tolerance and CVD. In the Nurses' Health Study, among women who reported having type 2 diabetes, the women who spent at least 4 hours per week performing moderate (including walking) or vigorous exercise had a 40% lower risk of developing CVD (including coronary heart disease [CHD] and stroke) than those who did not. In another study of 2,896 adults with diabetes, those who walked for at least 2 hours per week had lower CV mortality rates compared to inactive individuals (hazard ratio [HR] 0.66, 95% CI 0.45–0.96) (23). Rates were even lower for those who walked 3 to 4 hours per week (HR 0.47, 95% CI 0.24–0.91).
Nutrition therapy
The CVD event risk reduction benefits of a Mediterranean style diet are well documented (see Nutrition Therapy chapter, p. S64) and may be related to anti-inflammatory and antioxidative effects. The PREDIMED (Prevencion con Dieta Mediterranea trial) randomized nearly 7,500 participants at high CV risk to a Mediterranean diet supplemented with extra-virgin olive oil or mixed nuts, or to a control diet. About 50% of participants had type 2 diabetes. The trial was stopped early after a 30% reduction in the primary composite outcome of CV death, MI or stroke was observed with the Mediterranean diet. People with existing diabetes (n=3,614) had results similar to the main trial population.
Weight management
No randomized prospective trials, including the Look AHEAD trial discussed above, have shown a reduction in major CV endpoints or mortality with weight loss in people with diabetes and obesity (22) (see Physical Activity and Diabetes chapter, p. S54).
Glycemic control
The Diabetes Control and Complications Trial (DCCT) in type 1 diabetes (24), the Kumamoto trial (25), and the United Kingdom Prospective Diabetes Study (UKPDS) (26,27) in type 2 diabetes demonstrated that improved glycemic control significantly reduced the risk of microvascular complications, but had no significant effect on CV outcomes. Subsequent observational data from long-term follow up after termination of randomization periods of both the DCCT and UKPDS cohorts showed a persistence of significant microvascular benefits and also demonstrated an emergence of beneficial effect on CV outcomes attributed to intensive glycemic control (28,29) (see Targets for Glycemic Control chapter, p. S42).
Three major randomized controlled trials—the Action to Control Cardiovascular Risk in Diabetes (ACCORD) (30), Action in Diabetes and Vascular Disease: Preterax and Diamicron MR Controlled Evaluation (ADVANCE) (31,32), and Veterans Affairs Diabetes Trial (VADT) (33,34)—examined the effect of intensive glycemic control on middle-aged or older (mean age 60 to 68 years) participants with established type 2 diabetes for 8 to 11 years, with either CVD or multiple CVD risk factors. These studies compared intensive glycemic control with an A1C of 6.4% to 6.9% vs. 7.0% to 8.4% in the standard glycemic control cohort. No benefit on CV outcomes was seen in any of the 3 studies. The ACCORD trial was stopped early because of a 22% increase in all-cause mortality (HR 1.22, 95% CI 1.01-1.46) driven predominantly by CV mortality (30). The reasons for the increased mortality associated with intensive glycemic control are unclear (see Targets for Glycemic Control chapter, p. S42).
A retrospective analyses of data from the ADVANCE trial suggests that visit-to-visit variability in A1C and fasting plasma glucose predicted future CV events, microvascular events and all-cause mortality independent of CVD risk factors (35). Glycemic variability has been linked to mitochondrial superoxide overproduction, and oxidative stress is a key driver of atherosclerotic disease development and progression (36–39). In addition, glycemic variability has been linked to increases in inflammatory cytokines and increased macrophage and monocyte adhesion to the vascular endothelium, also promoting the development and progression of atherosclerosis (35–39). In one cohort study of >5,000 people with type 2 diabetes, time-dependent variation of fasting glycemia was a strong predictor of all-cause and CV mortality (40) (see Targets for Glycemic Control chapter, p. S42).
Antihyperglycemic agents
Based on controversies regarding rosiglitazone, in 2008, the United States Food and Drug Administration (FDA) required that all new antidiabetic therapies undergo evaluation for CV safety at the time of approval. Since then, several trials have reported evaluations of dipeptidyl peptidase (DPP)-4 inhibitors (41–43), glucagon-like polypeptide (GLP)-1 receptor agonists (44–46), sodium-glucose cotransporter (SGLT)-2 inhibitors (47,48) and insulin (49); and many other trials are underway. These studies were done in high-risk people with diabetes with either established CVD or multiple CV risk factors, and are discussed in detail in the Pharmacologic Glycemic Management of Type 2 Diabetes in Adults chapter, p. S88).
In general, the DPP-4 inhibitors studied thus far have demonstrated non-inferiority/safety for MACE. The exception was the Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus (SAVOR-TIMI 53) trial with saxagliptin, where there was an observed increase in the risk of hospitalization for heart failure (41).
In the Empagliflozin Cardiovascular Outcome Event (EMPA-REG OUTCOME) trial, which included 7,020 people with type 2 diabetes and clinical CVD, the SGLT2 inhibitor, empagliflozin, demonstrated a significant reduction in MACE and heart failure hospitalizations, driven by a marked reduction in CV mortality and all-cause mortality (47). There was no heterogeneity observed between the doses of empagliflozin 10 mg or 25 mg, and therapy was generally very well tolerated. Although the exact mechanism(s) of benefit of empagliflozin remains unclear, the observed CVD risk reduction was driven by a reduction in hospitalization for heart failure and CV mortality and not via a reduction in fatal and non-fatal atherothrombotic events. In the Canagliflozin Cardiovascular Assessment (CANVAS) Study, which combined data from 2 trials involving a total of 10,142 participants with type 2 diabetes and high CV risk, the SGLT2 inhibitor, canagliflozin, demonstrated a reduction in MACE (48); however, there was an increase in the risk of lower limb amputations and fractures in the canagliflozin group.
The Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial enrolled 9,340 participants with longstanding type 2 diabetes (45). The majority of participants (81%) were ≥50 years of age on pre-existing antihyperglycemic therapy with at least 1 CV condition (coronary heart disease, cerebrovascular disease, peripheral arterial disease, heart failure, or stage 3 or higher chronic kidney disease [CKD]). Over a median follow up of 3.8 years, fewer participants in the GLP-1 receptor agonist, liraglutide, arm compared to placebo had the primary endpoint of CV death, nonfatal MI or nonfatal stroke (13.0% vs. 14.9%, respectively; HR 0.87, 95% CI 0.78–0.97), fulfilling the statistical criteria for both noninferiority (p<0.001) and superiority (p=0.01). The Trial to Evaluate Cardiovascular and Other Long-term Outcomes with Semaglutide in Subjects with Type 2 Diabetes (SUSTAIN-6) enrolled 3,297 participants with a mean duration of type 2 diabetes of 13.9 years (46). At baseline, 83% had established CVD or stage 3 or higher CKD. After a median follow up of 2.1 years, the primary composite outcome of CV death, nonfatal MI or nonfatal stroke occurred in 6.6% of participants treated with the GLP-1 receptor agonist, semaglutide, and 8.9% of participants treated with placebo (HR 0.74, 95% CI 0.58–0.95), fulfilling statistical criteria for noninferiority (p<0.001); a non-pre-specified test for superiority was also significant (p=0.02).
The CV safety of sulfonylureas and meglitinides is uncertain (50,51). In a meta-analysis of 115 trials (of at least 6 months duration) comparing sulfonylureas with an active comparator in people with type 2 diabetes, there was no difference in the incidence of MACE, although overall mortality (but not CV mortality) was increased (odds ratio [OR] 1.22, 95% CI 1.01–1.49) (52). In a subsequent meta-analysis of 47 trials (of at least 1-year duration) comparing second-generation sulfonylureas (gliclazide, glimepiride) with diet, placebo or an active comparator, sulfonylureas were not associated with an increased risk of overall mortality, CV mortality, MI or stroke (53). Trials comparing metformin and sulfonylurea have suggested higher rates of cardiac events with sulfonylureas than metformin (54–56); however, it is not known whether the increase in CV risk is due to CV toxicity from sulfonylureas or from the possibly protective effects of metformin.
Blood pressure control
Hypertension is very common in persons with diabetes. Recommended BP targets and pharmacological therapies are discussed in the Treatment of Hypertension chapter, p. S186.
Antiplatelet therapy
Primary prevention
Platelets play a pivotal role in the development of atherosclerosis and vascular thrombosis. As people with diabetes have increased in vitro platelet reactivity and aggregation, they might be expected to have enhanced benefit from platelet inhibition with agents, such as acetylsalicylic acid (ASA). However, in vitro tests of platelet aggregation suggest that people with diabetes have platelets that are more likely to be resistant to the inhibitory effect of ASA (57,58). Thus, despite the proven advantages of ASA therapy in people with established CVD, the evidence for benefits of ASA therapy for the primary prevention of CVD events in persons with diabetes is less robust (59). More recently, a subgroup analysis of the Japanese Prevention of Atherosclerosis (JPAD) trial of ASA in the primary prevention of CVD events in diabetes has suggested that persons with diabetes and an elevated C-reactive protein level may benefit from ASA (60).
Pooled estimates suggest that, for primary prevention of CVD events in people with diabetes, ASA results in no reduction of MI and stroke, but an important increase in gastrointestinal hemorrhage (61–64).
Despite a plethora of data, there remains uncertainty about the use of ASA in the primary prevention of CVD events in persons with diabetes, and its routine use in primary CVD event prevention is not recommended. However, some people with multiple CV risk factors and evidence of vascular inflammation, as reflected by C-reactive protein levels, may cross the risk-benefit threshold in which the potential benefits justify the potential increase in hemorrhagic events.
Existing evidence suggests that some people with diabetes may be resistant to the effects of ASA for a number of reasons. The Study Comparing Cardiovascular Effects of Ticagrelor vs. Placebo in Patients with Type 2 Diabetes Mellitus (THEMIS) is currently underway and is examining the role of the adenosine receptor antagonist, ticagrelor, in primary prevention of MACE in people with type 2 diabetes (65).
Secondary prevention
ASA has been shown to reduce CVD events in people with and without diabetes and established CVD (66). The clinical trial evidence, as reflected in the 2011 Canadian Cardiovascular Society Guidelines on the “Use of Antiplatelet Therapy in the Outpatient Setting”, supports the use of ASA 75 mg to 162 mg daily for the secondary prevention of CVD events in those with diabetes (67).
Clopidogrel 75 mg may be used in people unable to tolerate ASA. The Clopidogrel vs. Aspirin in Patients at Risk of Ischaemic Events (CAPRIE) trial found that clopidogrel had a modest and marginally significant advantage over aspirin for the prevention of stroke, MI, and vascular disease in 19,185 participants with a recent stroke, MI or peripheral artery disease (annual event rate 5.3% vs. 5.8%) although the study population was not specific for people with diabetes (68).
In addition, there is evidence from the Prevention of Cardiovascular Events in Patients with Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin-Thrombolysis in Myocardial Infarction 54 (PEGASUS-TIMI 54) trial to indicate that people with diabetes and a prior MI may experience a reduction in MACE from dual antiplatelet therapy, with both ASA and ticagrelor, when this therapy is extended more than 1 year beyond the original acute coronary event (69); however, increased major bleeding was observed (HR 2.56, 95% Cl 1.52–4.33, p=0.0004).
Renin angiotensin aldosterone system (RAAS) inhibition
The benefit of angiotensin-converting enzyme (ACE) inhibition for CV protection with ramipril 10 mg daily was demonstrated by the Heart Outcomes Prevention Evaluation (HOPE) trial in participants with and without diabetes (70). It was also shown in the Micro-HOPE subset analysis of participants with diabetes, which enrolled individuals with diabetes, aged ≥55 years, with 1 other CV risk factor (total cholesterol >5.2 mmol/L, HDL-C <0.9 mmol/L, hypertension, microalbuminuria or smoking) or established CVD (71). In participants with diabetes enrolled in the European Trial on Reduction of Cardiac Events with Perindopril in Stable Coronary Artery Disease (EUROPA) study, the benefits from perindopril 8 mg daily were similar to those observed in the overall group; however, in this subgroup, the sample size was too small to show a statistically significant benefit (72). The Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial (ONTARGET) indicated similar CV protective effect from the angiotensin receptor blocker (ARB) telmisartan 80 mg daily as the ACE inhibitor ramipril 10 mg daily in a subset of participants with diabetes (73).
Whether the benefits of ACE inhibition result from a reduction in BP or from an unique CV benefit remains controversial. The benefits of ACE inhibition in both the HOPE and EUROPA trials were observed in individuals with or without a history of hypertension, and in those with higher and lower BP readings (70,74). In the HOPE study, after adjustment for the changes in systolic (2.4 mmHg) and diastolic (1.0 mmHg) BPs, ramipril still lowered the risk of the combined primary outcome by 25% (95% Cl 12–36, p=0.0004) (71).
One meta-analysis demonstrated a significant reduction in all-cause mortality, CV mortality and major CV events with ACE inhibitors in people with diabetes (75). Twenty-three of 35 identified trials compared ACE inhibitors with placebo (n=11) or active drugs (n=12) (32,827 participants) and 13 compared ARBs with active drugs (n=3) or placebo (n=10) (23,867 participants); the vast majority of studies were performed in participants with hypertension. When compared with controls (placebo/active treatment), ACE inhibitors significantly reduced the risk of all-cause mortality by 13% (risk reduction [RR] 0.87, 95% CI 0.78–0.98), CV deaths by 17% (RR 0.83, 95% Cl 0.70–0.99) and major CV events by 14% (RR 0.86, 95% Cl 0.77–0.95). Treatment with ARBs did not significantly affect all-cause mortality (RR 0.94, 95% CI 0.82–1.08), CV death rate (RR 1.21, 95% CI 0.81–1.80) and major CV events (RR 0.94, 95% CI 0.85–1.01).
In contrast, a recent meta-analyses of 19 randomized controlled trials (25,414 participants) that compared the use of ACE inhibitors and ARBs to other antihypertensive agents in people with diabetes found no difference in CV outcomes (76). When compared with other antihypertensive agents, ACE inhibitors and ARBs were associated with a similar risk of death (RR 0.99, 95% CI 0.93–1.05), CV death (RR 1.02, 95% CI 0.83–1.24) and MI (RR 0.87, 95% CI 0.64–1.18). There was also no difference in the hard renal outcome of ESRD (RR 0.99, 95% CI 0.78–1.28). Although the authors acknowledged that while doubling of creatinine is a stringent and commonly used endpoint and even though there are trials that have shown benefit of renin angiotensin aldosterone system (RAAS) on this outcome, doubling of creatinine was not consistently reported in the trials reviewed and was not examined explicitly in their analysis.
There is emerging uncertainty as to whether the use of RAAS blockade in people with diabetes, but without a history of hypertension or CV risk factors, derive a CV event reduction benefit from being placed on RAAS inhibition. Given the significant differences in the clinical trial protocols, the specific patient populations, the durations of the interventions, the conflicting findings between renal outcomes and CV outcomes, and the durations of the trial follow ups, it is difficult to state emphatically whether or not RAAS inhibition, independent of the presence of hypertension, is a benefit in all people with diabetes. The Vascular Protection in People with Diabetes chapter in the Canadian Diabetes Association 2013 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canadarecommended that all people with diabetes ≥55 years of age be started on an ACE inhibitor or ARB, at doses that have demonstrated vascular protection, even in the absence of a CV risk factor or end organ damage (i.e. albuminuria, retinopathy, left ventricular hypertrophy) (77). This Grade D, Consensus recommendation has been eliminated from the 2018 guidelines as no studies have clearly demonstrated CV benefit for this specific lower-risk population. However, it is important to recognize that the overwhelming majority of people with diabetes have additional compelling indications to be on RAAS inhibitors by age 55 years, almost always require multiple medications to achieve BP targets and almost always have suboptimal BP control. Therefore, the clinical likelihood that people with diabetes will end up on some form of RAAS inhibition remains extremely high. While the recommendation to use RAAS therapy in all adults ≥55 years has been removed, we strongly encourage clinicians to regularly evaluate CV risk in all persons with diabetes to ensure people with diabetes who would benefit from RAAS inhibition are identified and treated appropriately.
Lipid-modifying therapies
There is clinical trial evidence of the benefits of statin therapy for primary prevention in people with diabetes at ages prior to achieving a high proximate 10-year CVD risk. The Heart Protection Study (HPS) enrolled 5,963 individuals from age 40 years with diabetes, of whom 49% had no evidence of CVD (78). CV events were reduced by 22% (95% CI 13–30) in the participants with diabetes receiving simvastatin 40 mg daily for the 5-year treatment period (79). The same relative benefit was observed in participants with or without evidence of CVD. In the 615 participants with type 1 diabetes, there was a similar (although not statistically significant) risk reduction as observed in the 5,438 participants with type 2 diabetes. The Collaborative Atorvastatin Diabetes Study (CARDS) included 2,838 participants with diabetes, 1 CV risk factor and age >40 years (80). They were treated for an average of 3.9 years with either atorvastatin 10 mg daily or placebo. CV events were reduced by 37% (95% CI −52% to -17%, p=0.001) by atorvastatin compared to placebo, with a 36% reduction of acute coronary heart disease, a 31% reduction of coronary revascularization and a 48% reduction of stroke. There was a strong trend toward a 27% reduction in all-cause mortality (95% CI −48% to 1%, p=0.059). Consequently, both the HPS and CARDS studies provided evidence supporting the use of statin therapy for all people with diabetes ≥40 years of age with or without 1 CV risk factor. The CARDS study concluded with the statement: “The debate about whether all patients with type 2 diabetes warrant statin treatment should now focus on whether any patients can reliably be identified as being at sufficiently low risk for this safe and efficacious treatment to be withheld” (80).
As a direct reflection of the impact of diabetes on lifetime risk for CVD, increased vascular aging, premature development of CVD, shorter life expectancy for the individual with diabetes, poor predictive value of current risk models and studies demonstrating benefit of lipid lowering in people with diabetes, the current guidelines recommend statin therapy for primary CVD prevention for all people with diabetes ≥40 years of age. The guidelines also continue to support the use of statins in secondary prevention in those with evidence of end organ damage (CVD, microvascular disease, particularly albuminuria). In addition, there are other circumstances, not specific to diabetes, that may warrant statin therapy for a particular individual based on the 2016 Canadian Cardiovascular Society (CCS) Guidelines for the Management of Dyslipidemia(81).
LDL-C reduction should aim to achieve targets recommended in the current guidelines, and statins should be prescribed up to the maximally tolerated and approved dose. However, the use of other lipid-lowering agents in addition to statins may be necessary in some patients to achieve LDL-C goals (see Dyslipidemia chapter, p. S178). The IMProved Reduction of Outcomes: Vytorin Efficacy International trial (IMPROVE-IT) showed that the addition of ezetimibe to simvastatin in participants with recent acute coronary syndrome imparted an incremental CVD event benefit compared to use of simvastatin alone and the magnitude of the event reduction was commensurate with the degree of additional LDL-C lowering imparted by ezetimibe. The mean LDL-C in the simvastatin plus ezetimibe arm was 1.4 mmol/L and 1.8 mmol/L in the simvastatin-treated cohort. The event reductions were particularly evident in people with type 2 diabetes (82). Whether this is a specific effect of this drug combination or simply a reflection of the additional reduction in LDL-C remains unknown; however, existing data point strongly to the additional reduction in LDL-C as being significantly beneficial in people with diabetes.
Most recently, proprotein convertase subtilisin/kexin type 9 (PCSK-9) inhibitors have been shown to add clinically significant LDL-C lowering when added to standard therapy (83,84). The Further Cardiovascular Outcomes Research with PCSK9 Inhibition in Subjects with Elevated Risk (FOURIER) study of evolocumab vs. placebo in stable CAD and CVD participants receiving moderate to high-dose statin therapy, demonstrated a 15% reduction in MACE (CV death, MI, stroke, hospital admission for unstable angina or coronary revascularization) (85). The study included more than 11,000 participants with diabetes and a sub-analysis revealed that risk reductions in participants with or without diabetes were similar (HR 0.83, 95% CI 0.75–0.93, p=0⋅0008 for participants with diabetes and 0.87, 95% Cl 0⋅79–0⋅96, p=0⋅0052 for participants without diabetes) (86).
7. Other Relevant Guidelines
- Targets for Glycemic Control, p. S42
- Physical Activity and Diabetes, p. S54
- Nutrition Therapy, p. S64
- Pharmacologic Glycemic Management of Type 2 Diabetes in Adults, p. S88
- Weight Management in Diabetes, p. S124
- Dyslipidemia, p. S178
- Treatment of Hypertension, p. S186
Literature Review Flow Diagram for Chapter 23: Cardiovascular Protection in People with Diabetes
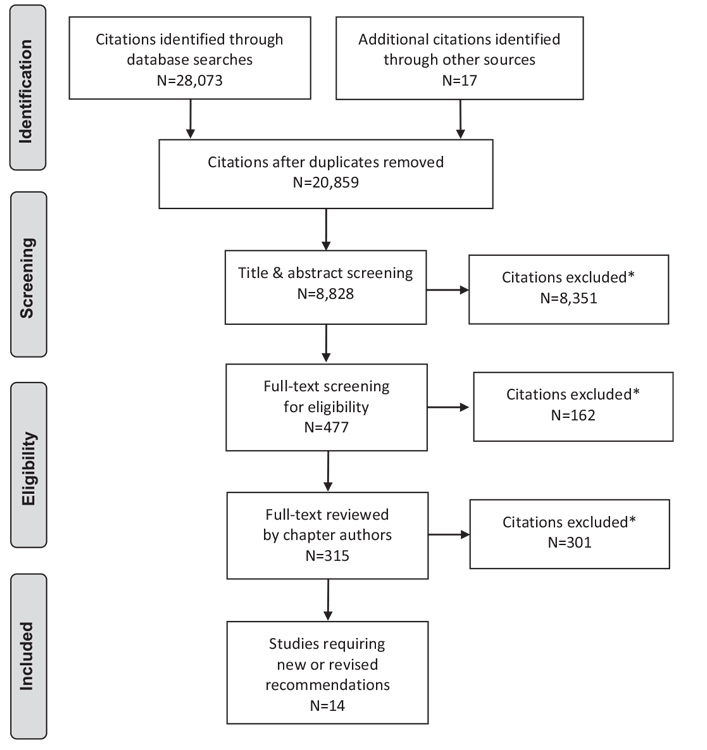
*Excluded based on: population, intervention/exposure, comparator/control or study design.
From: Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med 6(6): e1000097. doi:10.1371/journal.pmed1000097 (89).
For more information, visit www.prisma-statement.org.
8. Author Disclosures
Dr. Stone reports personal fees from AstraZeneca, Amgen, Bayer, Boehringer Ingelheim, Lilly, Novo Nordisk, Sanofi, and Servier, outside the submitted work. Dr. Houlden reports grants from Boehringer Ingelheim, Novo Nordisk, and Eli Lilly, outside the submitted work. Dr. Lin reports personal fees from AstraZeneca, Boehringer Ingelheim, Bayer, Eli Lilly, Merck, Sanofi, Amgen, Janssen, Novartis, Servier, Mylan, and Astellas, outside the submitted work. Dr. Udell has consultant or advisory relationships with Amgen, Boehringer Ingelheim, Janssen, Merck, Novartis, and Sanofi; has received research support from AstraZeneca and Novartis; and has received honoraria for sponsored lectures from Boehringer Ingelheim and Janssen. Dr. Verma reports grants and personal fees from Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, and Valeant; personal fees from Bayer, Janssen, Merck, Novartis, Novo Nordisk, and Sanofi, outside the submitted work. Dr. Verma also reports personal fees from Abbott and grants and personal fees from Bristol Myers Squibb-Pfizer.
Resources
-
Interactive tools
Individualizing your patient’s A1C target
For patients with type 1 and type 2 diabetesPatient specific A1C target recommendation
-
Interactive tools
Pharmacotherapy for type 2 diabetes
Individualize by agent and patient characteristics
-
Interactive tools
Reducing vascular risk
Does this patient require vascular protective medication?To be used for vascular protection, even if the baseline blood pressure or LDL-C is at...
-
Interactive tools
-
PDF
2018 Clinical Practice Guidelines Quick Reference Guide
Includes:Screening @ Diagnosis Quick Reference3 Quick Questions to Ask Patients To Meet Their Goals Quick ReferenceIndividualized Goal Setting...
-
PDF
-
Content
ABCDES of diabetes care quick reference guide
GUIDELINE TARGET (or personalized goal)AA1C targetsA1C ≤7% If on insulin or insulin secretagogue, assess for hypoglycemia and ensure driving...
-
Content
ABCDESSS of staying healthy with diabetes
Ask your health-care team about your:AA1C @ target is usually 7% or lessBBP @ aim for less than 130/80 mmHgCCholesterol @ LDL cholesterol should be...
Recommendations
-
All individuals with diabetes should follow a comprehensive, multifaceted approach to reduce CV risk, including:
- A1C ≤7.0% implemented early in the course of diabetes [Grade C, Level 3 (28,29)]
- Systolic BP of <130 mmHg [Grade C, Level 3 (87)] and diastolic BP of <80 mmHg [Grade B, Level 1 (88)] (see Treatment of Hypertension chapter, p. S186)
- Additional vascular-protective medications in the majority of adults with diabetes (see recommendations below) [Grade A, Level 1 (3,4) for those with type 2 diabetes age >40 years with albuminuria; Grade D, Consensus for those with type 1 diabetes]
- Achievement and maintenance of healthy weight goals [Grade D, Consensus]
- Healthy eating (see Nutrition Therapy chapter, p. S64 for specific dietary recommendations)
- Regular physical activity [Grade D, Consensus] (see Physical Activity chapter, p. S54)
- Smoking cessation [Grade C, Level 3 (20)].
-
Statin therapy should be used to reduce CV risk in adults with type 1 or type 2 diabetes with any of the following features:
- Clinical CVD [Grade A, Level 1 (79)]
- Age ≥40 years [Grade A, Level 1 (79,80), for type 2 diabetes; Grade D, Consensus for type 1 diabetes]
-
Age <40 years and 1 of the following:
- Diabetes duration >15 years and age >30 years [Grade D, Consensus]
- Microvascular complications [Grade D, Consensus]
- Warrants therapy based on the presence of other CV risk factors according to the 2016 Canadian Cardiovascular Society Guidelines for the Diagnosis and Treatment of Dyslipidemia (81)[Grade D, Consensus].
- For individuals not at LDL-C goal despite statin therapy, a combination of statin therapy with second-line agents may be used to achieve the goal and the agent used should be selected based upon the size of the existing gap to LDL-C goal [Grade D, Consensus]. Generally, ezetimibe should be considered [Grade D, Consensus]. In people with diabetes who also have concomitant clinical CVD, ezetimibe or evolocumab may be used to further reduce major adverse cardiac events [Grade A, Level 1 (82) for ezetimibe, Grade A, Level 1 (85) for evolocumab], and they should also be considered in those with concomitant familial hypercholesterolemia [Grade D, Consensus for ezetimibe and PCSK9 inhibitor].
- ACE inhibitor or ARB, at doses that have demonstrated vascular protection, should be used to reduce CV risk in adults with type 1 or type 2 diabetes with any of the following:
- In people with established CVD, low-dose ASA therapy (81–162 mg) should be used to prevent CV events [Grade B, Level 2 (66)].
- ASA should not be used routinely for the primary prevention of CVD events in people with diabetes [Grade A, Level 1A (62–64)]. ASA may be used in the presence of additional CV risk factors [Grade D, Consensus].
- Clopidogrel 75 mg may be used in people unable to tolerate ASA [Grade D, Consensus].
- In adults with type 2 diabetes with clinical CVD in whom glycemic targets are not achieved with existing antihyperglycemic medication(s) and with an eGFR > 30mL/min/1.73 m2, an antihyperglycemic agent with demonstrated CV outcome benefit should be added to reduce the risk of major CV events [Grade A, Level 1A (47)for empagliflozin; Grade A, Level 1A for liraglutide (45); Grade C, Level 2 for canagliflozin (48) (see Pharmacologic Glycemic Management of Type 2 Diabetes in Adults chapter, p. S88).
Note: Among women with childbearing potential, ACE inhibitors, ARBs or statins should only be used if there is reliable contraception.
Abbreviations:
A1C, glycated hemoglobin; BG, blood glucose; BP, blood pressure; CAD, coronary artery disease; CI, confidence interval; CKD, chronic kidney disease; CV, cardiovascular; CVD, cardiovascular disease; CKD, chronic kidney disease; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; MACE, major cardiovascular events; NNT, number needed to treat; RAAS, renin angiotensin aldosterone system; RR, relative risk.
References
- Haffner SM, Lehto S, Ronnemaa T, et al. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 1998;339:229–34.
- Stevens RJ, Kothari V, Adler AI, et al. The UKPDS risk engine: A model for the risk of coronary heart disease in type II diabetes (UKPDS 56). Clin Sci 2001;101:671–9.
- Gaede P, Vedel P, Larsen N, et al. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 2003;348:383–93.
- Gaede P, Lund-Andersen H, Parving HH, et al. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med 2008;358:580–91.
- Bittner V, Bertolet M, Barraza Felix R, et al. Comprehensive cardiovascular risk factor control improves survival: The BARI 2D trial. J AmColl Cardiol 2015;66:765–73.
- Booth GL, Kapral MK, Fung K, et al. Relation between age and cardiovascular disease in men and women with diabetes compared with non-diabetic people: A population-based retrospective cohort study. Lancet 2006;368:29–36.
- Stock S, Drabik A, Buscher G, et al. German diabetes management programs improve quality of care and curb costs. Health Aff (Millwood) 2010;29:2197–205.
- Bergner DW, Goldberger JJ. Diabetes mellitus and sudden cardiac death: What are the data? Cardiol J 2010;17:117–29.
- Guzder RN, Gatling W, Mullee MA, et al. Prognostic value of the Framingham cardiovascular risk equation and the UKPDS risk engine for coronary heart disease in newly diagnosed type 2 diabetes: Results froma United Kingdom study. Diabet Med 2005;22:554–62.
- Lloyd-Jones DM, Leip EP, Larson MG, et al. Prediction of lifetime risk for cardiovascular disease by risk factor burden at 50 years of age. Circulation 2006;113:791–8.
- Stone JA. Framing cardiovascular disease event risk prediction. Can J Cardiol 2011;27:171–3.
- Fox CS, Pencina MJ, Wilson PWF, et al. Lifetime risk of cardiovascular disease among individuals with and without diabetes stratified by obesity status in the Framingham Heart study. Diabetes Care 2008;31:1582–4.
- Dagenais GR, Pogue J, Fox K, et al. Angiotensin-converting-enzyme inhibitors in stable vascular disease without left ventricular systolic dysfunction or heart failure: A combined analysis of three trials. Lancet 2006;368:581–8.
- Zachary I, Mathur A, Yla-Herttuala S, et al. Vascular protection: A novel nonangiogenic cardiovascular role for vascular endothelial growth factor. Arterioscler Thromb Vasc Biol 2000;20:1512–20.
- Long GH, Cooper AJM, Wareham NJ, et al. Healthy behavior change and cardiovascularoutcomes innewly diagnosed type 2 diabetic patients: A cohort analysis of the addition-cambridge study. Diabetes Care 2014;37:1712–20.
- Tobe SW, Stone JA,Walker KM, et al. Canadian Cardiovascular Harmonized National Guidelines Endeavour (C-CHANGE): 2014 update. CMAJ 2014;186:1299–305.
- de Meirelles LR, Matsuura C, Resende Ade C, et al. Chronic exercise leads to antiaggregant, antioxidant and anti-inflammatory effects in heart failure patients. Eur J Prev Cardiol 2014;21:1225–32.
- Folsom AR, Szklo M, Stevens J, et al. A prospective study of coronary heart disease in relation to fasting insulin, glucose, and diabetes. The Atherosclerosis Risk in Communities (ARIC) study. Diabetes Care 1997;20:935–42.
- Yacoub R, Habib H, Lahdo A, et al. Association between smoking and chronic kidney disease: A case control study. BMC Public Health 2010;10:731.
- Chaturvedi N, Stevens L, Fuller JH. Which features of smoking determine mortality risk in former cigarette smokers with diabetes? The World Health Organization Multinational Study Group. Diabetes Care 1997;20:1266–72.
- Chudyk A, Petrella RJ. Effects of exercise on cardiovascular risk factors in type 2 diabetes: A meta-analysis. Diabetes Care 2011;34:1228–37.
- Look AHEAD Research Group, Wing RR, Bolin P, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med 2013;369:145–54.
- Gregg EW, Gerzoff RB, Caspersen CJ, et al. Relationship of walking to mortality among US adults with diabetes. Arch Intern Med 2003;163:1440–7.
- Diabetes Control and Complications Trial Research Group, Nathan DM, Genuth S, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993;329:977–86.
- Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: A randomized prospective 6-year study. Diabetes Res Clin Pract 1995;28:103–17.
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998;352:837–53.
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive bloodglucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998;352:854–65.
- Holman RR, Paul SK, Bethel MA, et al. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–89.
- Nathan DM, Cleary PA, Backlund JY, et al. Intensive diabetes treatment and cardiovascular disease in patientswith type 1 diabetes. N Engl JMed 2005;353:2643–53.
- Action to Control Cardiovascular Risk in Diabetes Study Group, Gerstein HC, Miller ME, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358:2545–59.
- ADVANCE Collaborative Group, Patel A, MacMahon S, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358:2560–72.
- Zoungas S, Chalmers J, Neal B, et al. Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med 2014;371:1392–406.
- Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009;360:129–39.
- Moritz T, Duckworth W, Abraira C. Veterans affairs diabetes trial–corrections. N Engl J Med 2009;361:1024–5.
- Hirakawa Y, Arima H, Zoungas S, et al. Impact of visit-to-visit glycemic variability on the risks of macrovascular and microvascular events and all-cause mortality in type 2 diabetes: The ADVANCE trial. Diabetes Care 2014;37:2359–65.
- Monnier L, Mas E, Ginet C, et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA 2006;295:1681–7.
- Brownlee M, Hirsch IB. Glycemic variability: A hemoglobin A1c-independent risk factor for diabetic complications. JAMA 2006;295:1707–8.
- Nishikawa T, Edelstein D, Du XL, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000;404:787–90.
- Ceriello A, Ihnat MA. “Glycaemic variability”: A new therapeutic challenge in diabetes and the critical care setting. Diabet Med 2010;27:862–7.
- Lin CC, Li CI, Yang SY, et al. Variation of fasting plasma glucose: A predictor of mortality in patients with type 2 diabetes. Am J Med 2012;125:416.e9–18.
- Scirica BM, Bhatt DL, Braunwald E, et al. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl JMed 2013;369:1317–26.
- Green JB, Bethel MA, Armstrong PW, et al. Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015;373:232–42.
- White WB, Cannon CP, Heller SR, et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med 2013;369:1327–35.
- Pfeffer MA, Claggett B, Diaz R, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 2015;373:2247–57.
- Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016;375:311–22.
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016;375:1834–44.
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117–28.
- Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017;377:644–57.
- Marso SP, McGuire DK, Zinman B, et al. Efficacy and safety of degludec versus glargine in type 2 diabetes. N Engl J Med 2017;377:723–32. Jun 12.
- Genuth S. Should sulfonylureas remain an acceptable first-line add-on to metformin therapy in patients with type 2 diabetes? No, it’s time to move on! Diabetes Care 2015;38:170–5.
- Abrahamson MJ. Should sulfonylureas remain an acceptable first-line add-on to metformin therapy in patients with type 2 diabetes? Yes, they continue to serve us well! Diabetes Care 2015;38:166–9.
- Monami M, Genovese S, Mannucci E. Cardiovascular safety of sulfonylureas: A meta-analysis of randomized clinical trials. Diabetes Obes Metab 2013;15:938–53.
- Varvaki Rados D, Catani Pinto L, Reck Remonti L, et al. The association between sulfonylurea use and all-cause and cardiovascular mortality: A meta-analysis with trial sequential analysis of randomized clinical trials. PLoS Med 2016;13:e1001992.
- Hong J, Zhang Y, Lai S, et al. Effects of metformin versus glipizide on cardiovascular outcomes in patients with type 2 diabetes and coronary artery disease. Diabetes Care 2013;36:1304–11.
- Simpson SH, Majumdar SR, Tsuyuki RT, et al. Dose-response relation between sulfonylurea drugs and mortality in type 2 diabetes mellitus: A populationbased cohort study. CMAJ 2006;174:169–74.
- Roumie CL, Hung AM, Greevy RA, et al. Comparative effectiveness of sulfonylurea and metformin monotherapy on cardiovascular events in type 2 diabetes mellitus: A cohort study. Ann Intern Med 2012;157:601–10.
- Angiolillo DJ, Suryadevara S. Aspirin and clopidogrel: Efficacy and resistance in diabetes mellitus. Best Pract Res Clin Endocrinol Metab 2009;23:375–88.
- Angiolillo DJ, Fernandez-Ortiz A, Bernardo E, et al. Influence of aspirin resistance on platelet function profiles in patients on long-term aspirin and clopidogrel after percutaneous coronary intervention. Am J Cardiol 2006;97:38–43.
- Ogawa H, Nakayama M, Morimoto T, et al. Low-dose aspirin for primary prevention of atherosclerotic events in patients with type 2 diabetes: A randomized controlled trial. JAMA 2008;300:2134–41.
- Soejima H, Ogawa H, Morimoto T, et al. Aspirin possibly reduces cerebrovascular events in type 2 diabetic patients with higher C-reactive protein level: Subanalysis from the JPAD trial. J Cardiol 2013;62:165–70.
- Canadian Diabetes Association Clinical Practice Guidelines Expert Committee, Stone JA, Fitchett D, et al. Vascular protection in people with diabetes. Can J Diabetes 2013;37:S100–4.
- Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: A systematic evidence review for the U.S. preventive services task force. Ann Intern Med 2016;164:804–13.
- De Berardis G, Sacco M, Strippoli GF, et al. Aspirin for primary prevention of cardiovascular events in people with diabetes: Meta-analysis of randomised controlled trials. BMJ 2009;339:b4531.
- Xie M, Shan Z, Zhang Y, et al. Aspirin for primary prevention of cardiovascular events: Meta-analysis of randomized controlled trials and subgroup analysis by sex and diabetes status. PLoS ONE 2014;9:e90286.
- A study comparing cardiovascular effects of ticagrelor versus placebo in patients with type 2 diabetes mellitus (THEMIS). ClinicalTrials.gov: AstraZeneca; 2017. Report No.: NCT01991795. https://clinicaltrials.gov/ct2/show/NCT01991795.
- Antithrombotic Trialists’ (ATT) Collaboration, Baigent C, Blackwell L, et al. Aspirin in the primary and secondary prevention of vascular disease: Collaborative metaanalysis of individual participant data from randomised trials. Lancet 2009;373:1849–60.
- Bell AD, Roussin A, Cartier R, et al. The use of antiplatelet therapy in the outpatient setting: Canadian Cardiovascular Society guidelines. Can J Cardiol 2011;27:S1–59.
- CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE steering committee. Lancet 1996;348:1329–39.
- Bhatt DL, Bonaca MP, Bansilal S, et al. Reduction in ischemic events with ticagrelor in diabetic patients with prior myocardial infarction in PEGASUS-TIMI 54. J Am Coll Cardiol 2016;67:2732–40.
- The Heart Outcomes Prevention Evaluation Study Investigators, Yusuf S, Sleight P, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. New Engl J Med 2000;342:145–53.
- Heart Outcomes Prevention Evaluation Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: Results of the HOPE study and MICRO-HOPE substudy. Lancet2000;355:253–9.
- Daly CA, Fox KM, Remme WJ, et al. The effect of perindopril on cardiovascular morbidity and mortality in patients with diabetes in the EUROPA study: Results from the PERSUADE substudy. Eur Heart J 2005;26:1369–78.
- ONTARGET Investigators, Yusuf S, Teo KK, et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med 2008;358:1547–59.
- Fox KM, EURopean trial On reduction of cardiac events with Perindopril in stable coronary Artery disease Investigators. Efficacy of perindopril in reduction of cardiovascular events among patients with stable coronary artery disease: Randomised, double-blind, placebo-controlled, multicentre trial (the EUROPA study). Lancet 2003;362:782–8.
- Cheng J, Zhang W, Zhang X, et al. Effect of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers on all-cause mortality, cardiovascular deaths, and cardiovascular events in patients with diabetes mellitus: A meta-analysis. JAA Intern Med 2014;174:773–85.
- Bangalore S, Fakheri R, Toklu B, et al. Diabetes mellitus as a compelling indication for use of renin angiotensin system blockers: Systematic review and metaanalysis of randomized trials. BMJ 2016;352:i438.
- Stone JA, Fitchett D, Grover S, et al. Vascular protection in people with diabetes. Can J Diabetes 2013;37(Suppl. 1):S100–4.
- Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: A randomised placebo-controlled trial. Lancet 2002;360:7–22.
- Collins R, Armitage J, Parish S, et al. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: A randomised placebo-controlled trial. Lancet 2003;361:2005–16.
- Colhoun HM, Betteridge DJ, Durrington PN, et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): Multicentre randomised placebocontrolled trial. Lancet 2004;364:685–96.
- Anderson TJ, Grégoire J, Pearson GJ, et al. 2016 Canadian cardiovascular society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in the adult. Can J Cardiol 2016;32:1263–82.
- Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015;372:2387–97.
- Sabatine MS, Giugliano RP, Wiviott SD, et al. Efficacy and safety of evolocumab in reducing lipids and cardiovascular events. New Engl J Med 2015;372:1500–9.
- Robinson JG, Farnier M, Krempf M, et al. Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. N Engl J Med 2015;372:1489–99.
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 2017;376:1713–22.
- Sabatine MS, Leiter LA, Wiviott SD, et al. Cardiovascular safety and efficacy of the PCSK9 inhibitor evolocumab in patients with and without diabetes and the effect of evolocumab on glycaemia and risk of new-onset diabetes: A prespecified analysis of the FOURIER randomised controlled trial. Lancet Diabetes Endocrinol 2017 (in press).
- Adler AI, Stratton IM, Neil HA, et al. Association of systolic blood pressure with macrovascular and microvascular complications of type 2 diabetes (UKPDS 36): Prospective observational study. BMJ 2000;321:412–19.
- Hansson L, Zanchetti A, Carruthers SG, et al. Effects of intensive bloodpressure lowering and low-dose aspirin in patients with hypertension: Principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study Group. Lancet 1998;351:1755–62.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med 2009;6: e1000097.