Overview
Diabetes Canada Clinical Practice Guidelines Expert Committee
Denice S. Feig MD, FRCPC, Howard Berger MD, Lois Donovan MD, FRCPC, Ariane Godbout MD, FRCPC, Tina Kader MD, FRCPC, Erin Keely MD, FRCPC, Rema Sanghera MA, RD
Anchored List of chapter sections
- Key Messages
- Key Messages for Women with Diabetes Who are Pregnant or Planning a Pregnancy
- Introduction
- Pre-Existing Diabetes (Type 1 and Type 2) in Pregnancy
- Management
- GDM
- Obstetrical Considerations in Women with Gestational Diabetes (See Section Entitled ‘Obstetrical Considerations in Women with Pre-Existing Diabetes and Gestational Diabetes’)
- Other Relevant Guidelines
- Author Disclosures
1. Key Messages
Pre-Existing Diabetes
Preconception and During Pregnancy
- All women with pre-existing type 1 or type 2 diabetes should receive preconception care to optimize glycemic control, assess for complications, review medications and begin folic acid supplementation.
- Effective contraception should be provided until the woman is ready for pregnancy.
- Care by an interprofessional diabetes health-care team composed of a diabetes nurse educator, dietitian, obstetrician and endocrinologist/internist with expertise in diabetes, both prior to conception and during pregnancy, has been shown to minimize maternal and fetal risks in women with pre-existing type 1 and type 2 diabetes.
- Women should aim for a glycated hemoglobin (A1C) of ≤7.0% (ideally ≤6.5% if possible) when planning pregnancy, or ≤6.5% (ideally ≤6.1% if possible) during pregnancy.
- Women should consider the use of the continuous glucose monitor during pregnancy to improve glycemic control and neonatal outcomes.
Postpartum
- All women should be given information regarding the benefits of breastfeeding, effective birth control and the importance of planning another pregnancy.
Gestational Diabetes Mellitus
During Pregnancy
- Untreated gestational diabetes leads to increased maternal and perinatal morbidity. Treatment reduces these adverse pregnancy outcomes.
- In women at high risk of undiagnosed type 2 diabetes, early screening (<20 weeks) with an A1C should be done to identify women with potentially overt diabetes to guide fetal surveillance and early maternal treatment, including self-monitoring of blood glucose, interventions that promote healthy behaviours and healthy weight gain.
- The diagnostic criteria for gestational diabetes (GDM) remain controversial; however, these guidelines identify a “preferred” and an “alternate” screening approach. The preferred approach is an initial 50 g glucose challenge test, followed, if abnormal, with a 75 g oral glucose tolerance test. A diagnosis of GDM is made if one plasma glucose value is abnormal (i.e. fasting ≥5.3 mmol/L, 1 hour ≥10.6 mmol/L, 2 hours ≥9.0 mmol/L). The alternate approach is a 1-step approach of a 75 g oral glucose tolerance test. A diagnosis of GDM is made if one plasma glucose value is abnormal (i.e. fasting ≥5.1 mmol/L, 1 hour ≥10.0 mmol/L, 2 hours ≥8.5 mmol/L).
- First-line therapy consists of diet and physical activity. If glycemic targets are not met, insulin or metformin can then be used.
Postpartum
- Women with gestational diabetes should be encouraged to breastfeed immediately after birth and for a minimum of 4 months to prevent neonatal hypoglycemia, childhood obesity, and diabetes for both the mother and child.
- Women should be screened for diabetes between 6 weeks and 6 months postpartum, with a 75 g oral glucose tolerance test and be given ongoing education regarding strategies to reduce the risk of developing type 2 diabetes.
2. Key Messages for Women with Diabetes Who are Pregnant or Planning a Pregnancy
Pre-Existing Diabetes
- The key to a healthy pregnancy for a woman with diabetes is keeping blood glucose levels in the target range—both before she is pregnant and during her pregnancy.
- Poorly controlled diabetes in a pregnant woman with type 1 or type 2 diabetes increases her risk of miscarrying, having a baby born with a malformation and having a stillborn.
-
Women with type 1 or type 2 diabetes should discuss pregnancy plans with their diabetes health-care team to:
- Review blood glucose targets
- Assess general health and status of any diabetes-related complications
- Aim for optimal weight and, if overweight, start weight loss before pregnancy with healthy eating
- Review medications
- Start folic acid supplementation (1.0 mg daily)
- Ensure appropriate vaccinations have occurred.
Gestational Diabetes
- Between 3% to 20% of pregnant women develop gestational diabetes, depending on their risk factors
-
Risk Factors include:
-
Being:
- 35 years of age or older
- from a high-risk group (African, Arab, Asian, Hispanic, Indigenous, or South Asian)
-
Using:
- Corticosteroid medication
-
Having:
- Obesity (a body mass index greater than or equal to 30 kg/m2)
- Prediabetes
- Gestational diabetes in a previous pregnancy
- Given birth to a baby that weighed more than 4 kg
- A parent, brother or sister with type 2 diabetes
- Polycystic ovary syndrome or acanthosis nigricans (darkened patches of skin).
-
Being:
- All pregnant women without known pre-existing diabetes should be screened for gestational diabetes between 24 to 28 weeks of pregnancy
-
If you were diagnosed with gestational diabetes during your pregnancy, it is important to:
- Breastfeed immediately after birth and for a minimum of 4 months in order to prevent hypoglycemia in your newborn, obesity in childhood, and diabetes for both you and your child
- Reduce your weight, targeting a normal body mass index in order to reduce your risk of gestational diabetes in the next pregnancy and developing type 2 diabetes
-
Be screened for type 2 diabetes after your pregnancy:
- within 6 weeks to 6 months of giving birth
- before planning another pregnancy
- every 3 years (or more often depending on your risk factors).
3. Introduction
This chapter discusses pregnancy in both pre-existing diabetes (type 1 and type 2 diabetes diagnosed prior to pregnancy), overt diabetes diagnosed early in pregnancy and gestational diabetes (GDM or glucose intolerance first recognized in pregnancy). Some management principles are common to all types of diabetes.
4. Pre-Existing Diabetes (Type 1 and Type 2) in Pregnancy
The term “pre-existing diabetes in pregnancy” refers to diabetes diagnosed before pregnancy. The prevalence of pre-existing diabetes has increased in the past decade (1) primarily as a result of the increase in type 2 diabetes (2). Studies of women with pre-existing diabetes show higher rates of complications compared to the general population, including perinatal mortality, congenital malformations, hypertension, preterm delivery, large-for-gestational-age (LGA) infants, caesarean delivery and other neonatal morbidities (1,3–5).
Preconception care
Preconception care improves maternal and fetal outcomes in women with pre-existing diabetes. This involves educating women about the importance of optimal glycemic control prior to pregnancy, discontinuing potentially harmful medications and achieving a health body weight. Hyperglycemia is teratogenic and if glycemic control is poor in the first few weeks of conception, the risk of congenital anomalies is increased. Women with diabetes should be helped to achieve optimal glycemic control preconception as this is associated with a reduction of congenital anomalies by 70% (6–9). However, even women who achieve a glycated hemoglobin (A1C) ≤7.0% preconception have an increased risk of complications compared to the general population. This may be caused, in part, by maternal obesity, especially in women with type 2 diabetes (10–13).
Preconception care should also include advice regarding folic acid supplementation. In 1 case-control study in the United States, women with diabetes who did not take folic acid containing vitamins were at a 3-fold higher rate of congenital anomalies compared to women with diabetes who did (14). There are no intervention trials to support folic acid doses greater than 1 mg for women with diabetes. Obesity, which is more common in women with type 2 diabetes, is associated with lower serum folate levels for the same intake, lower intake of folate rich foods and increased risk of neural tube defects independent of glycemic control (15–17). A higher dose of folic acid may be considered in women with obesity, although there is no clinical evidence that this higher dose reduces congenital anomalies. Measurement of red blood cell (RBC) folate may also be useful to guide adjustment of folic acid dosage in women with obesity or women who have had bariatric surgery.
A multifaceted preconception program that included patient information specialized clinics, electronic health records, online resources and local guidelines, increased folic acid use by 26%, improved glycemic control and decreased the risk of congenital malformations from 5% to 1.8% (9). Although receiving care at an interprofessional preconception clinic has been shown to be associated with improved pregnancy outcomes, approximately 50% of women do not receive such care (18,19). The following factors are associated with women with pre-existing diabetes being less likely to receive preconception care: overweight; younger age; smoking history; lower socioeconomic status; lower health literacy and/or poor relationship with their health-care provider (7,20–22). Additionally, some studies have shown that women with type 2 diabetes are less likely to receive preconception care compared to women with type 1 diabetes (19,23).
Assessment and management of complications
Retinopathy. Women with type 1 (24,25) and type 2 diabetes (26)should ideally have ophthalmological assessments before conception, during the first trimester, as needed during pregnancy, and within the first year postpartum (27,28). The risk of progression of retinopathy is increased with poor glycemic control during pregnancy, and progression may occur for up to 1 year postpartum (25,27). Additional risk factors for retinopathy progression include: chronic and pregnancy-induced hypertension, preeclampsia, more severe pre-existing diabetic retinopathy (24,29–31), and a greater decrease in A1C between the first and third trimester of pregnancy (32). Closer retinal surveillance is recommended for women with more severe pre-existing retinopathy, those with poor glycemic control or women with greater reductions in A1C during pregnancy (27,33). Laser photocoagulation for severe nonproliferative or proliferative retinopathy prior to pregnancy reduces the risk of visual impairment in pregnancy (34); if not performed prior to pregnancy, it is still considered safe to receive during pregnancy.
There is insufficient evidence to confirm safety or harm from the use of intravitreal antivascular endothelial growth factor (anti-VEGF) injections for diabetic macular edema or proliferative diabetic retinopathy during pregnancy (35). Potential side effects include hypertension, proteinuria, defective embryogenesis and fetal loss (36,37). It is not known if these medications cross the placenta or if they are secreted in breastmilk. Gestational timing of exposure needs to be considered in situations where potential benefit to the woman justifies the potential fetal risk. Until more safety information is available, we support the recommendations of others: a) to ensure a negative pregnancy test and contraception use during intravitreal anti-VEGF therapy, and b) to consider delaying conception for 3 months after the last intravitreal injection (38,39). Intravitreal anti-VEGF therapy in pregnancy should be avoided especially in the first trimester. Second and third trimester use should occur only if absolutely necessary after discussion of the potential risks and benefits. Diabetic macular edema may often regress after pregnancy without specific therapy. Data are lacking to guide treatment recommendations for diabetic macular edema during pregnancy.
One retrospective study of 193 women with type 1 diabetes, 63 with an active second-stage delivery (3 with proliferative diabetic retinopathy) found no impact of expulsive efforts in the active second stage of labour on retinopathy progression in women with stable retinopathy (40). Data from the Diabetes Control and Complications Trial (DCCT) has suggested that pregnancy does not affect the long-term outcome of mild-to-moderate retinopathy (27). More recently, preeclampsia and pregnancy-induced hypertension in women with type 1 diabetes has been shown to be associated with an increased risk of severe diabetic retinopathy later in life (41).
Hypertension. Women may have pre-existing hypertension or develop hypertension/preeclampsia during pregnancy. Women with type 1 and type 2 diabetes have a 40% to 45% incidence of hypertension complicating pregnancy (31). A systematic review of risk factors for preeclampsia demonstrated a 3.7 risk (relative risk [RR] 3.1 to 4.3) for the development of preeclampsia in women with pre-existing diabetes (42). Type 1 diabetes is more often associated with preeclampsia whereas type 2 diabetes is more often associated with chronic hypertension. In the general population, the risk of preeclampsia is highest in nulliparous women and lower in multiparous women. However, in women with type 1 diabetes, the risk of preeclampsia is similar in nulliparous and multiparous women (43). Other risk factors for hypertension, such as poor glycemic control in early pregnancy, are potentially modifiable. Some studies (44,45), but not all (46), have found that increased urinary protein excretion in early pregnancy is associated with an increased risk of hypertension.
Any type and degree of hypertension is associated with adverse outcomes. A large randomized controlled trial in pregnant women with nonproteinuric pre-existing or gestational hypertension (that included women with GDM) showed that targeting a diastolic blood pressure (BP) of 85 mmHg vs. 100 mmHg reduced neonatal respiratory complications, rates of severe maternal hypertension (i.e. >160/110 mmHg) and did not increase the incidence of small for gestational age (SGA) (47). Finally, a number of antihypertensive medications are safe and effective in pregnancy, including calcium channel blockers, labetalol and methyldopa.
Although there are no intervention trials for ASA prophylaxis for the prevention of preeclampsia specific to women with pre-existing diabetes, ASA prophylaxis started between 12 to 16 weeks of gestation is likely to be beneficial, given the evidence of benefit in other high-risk populations, (48).
Based on a meta-analysis and systematic review, calcium supplementation (of at least 1,000 mg/day) in high-risk populations, especially in those with low dietary calcium intake, may reduce preeclampsia rates by up to 40%, although evidence is limited (49).
Chronic kidney disease. Prior to conception, women should be screened for chronic kidney disease (CKD). Albuminuria and overt nephropathy are associated with increased risk of maternal and fetal complications (50–55). An estimated glomerular filtration rate (eGFR) should be used prior to pregnancy to determine risk of adverse outcomes. In 1 small study, women with poorer mean preconception creatinine clearance (CrCl) of 61 mL/min/1.73 m2 (range 37 to 73) showed a 36% lower creatinine clearance (CrCl) 3 months postpartum, whereas in women with a mean preconception CrCl of 80 mL/min/1.73 m2 (range 70 to 93), no deterioration in renal function was observed (56). However, inadequate BP control in pregnancy may account for this observed difference in this study.
During pregnancy, serum creatinine (not eGFR) should be used, as eGFR will underestimate GFR in pregnancy (57,58). Proteinuria increases during pregnancy, but, in women with a normal GFR, pregnancy has no adverse effects on long-term renal function as long as BP and blood glucose (BG) are well controlled (50–53,56,59,60). One small series found that women with serum creatinine >124 µmol/L at pregnancy onset had a greater than 40% chance of accelerated progression of diabetic nephropathy as a result of pregnancy (61). First trimester BP elevations and protein excretion are associated with delivery before 37 weeks, usually due to preeclampsia (62). Small cohort studies have suggested that antihypertensive therapy for BP >135/85 mmHg in women with diabetes and albuminuria during pregnancy may reduce the risk of preeclampsia and preterm delivery without adversely impacting other pregnancy outcomes (60,63,64).
There is conflicting information on whether first-trimester exposure to angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) is associated with an increased risk of congenital malformations (65,66). A meta-analysis, limited by small study size (n=786), demonstrated a significant risk ratio (relative risk [RR] 1.78, 95% confidence interval [CI] 1.07–2.94) of increased anomalies in infants exposed to first-trimester ACE inhibitors and ARBs compared to the general population (67). However, when the group exposed to ACE inhibitor/ARB was compared to a group of women who were exposed to other antihypertensives used in pregnancies, they were both associated with malformations with no statistically significant difference. Fetal exposure in the second and third trimesters is clearly associated with a fetal renin angiotensin aldosterone system (RAAS) blockade syndrome, which includes renal failure, oligohydramnios, hypotension, intrauterine growth restriction and death (68). The decision to discontinue an ACE inhibitor or ARB prior to pregnancy should be discussed with the woman and may depend on the indication for use and availability of an effective alternative medication. However, once a woman is pregnant, ACE inhibitors and ARBs should be discontinued.
Painful peripheral neuropathy management. As with all medications used in pregnancy, benefits need to be weighed against risk. In the relatively small number of reported pregnancies in which women were exposed to first trimester gabapentin monotherapy (n=294), no increased risk of congenital malformations was found (69,70). However, neonatal gabapentin withdrawal has been described with maternal oral gabapentin 600 mg 3 times daily throughout pregnancy (69).
Cardiovascular disease. Although rare, cardiovascular disease (CVD) can occur in women of reproductive age with diabetes. Myocardial infarct (MI) in pregnancy is associated with poor maternal and fetal outcomes (71,72). Women with known CVD should be evaluated and counselled about the significant risks associated with pregnancy. As well, statins and/or fibrates should be discontinued prior to pregnancy as they are not recommended for use during pregnancy.
5. Management
Care by an interprofessional diabetes health-care (DHC) team composed of diabetes nurse educators, dietitians, obstetricians and endocrinologists/internists with expertise in diabetes, both preconception and during pregnancy, has been shown to minimize maternal and fetal risks in women with diabetes (73–76)(see Organization of Care chapter, p. S27). An early working relationship should be established between the woman and the DHC team to optimize care, facilitate the planning of pregnancy, ensure adequate self-care practices and to discuss the need for social support during pregnancy.
Targets of glycemic control
Elevated BG levels have adverse effects on the fetus throughout pregnancy. At conception and during the first trimester, hyperglycemia increases the risk of fetal malformations and intrauterine fetal demise (77). Later in pregnancy, it increases the risk of macrosomia, fetal and infant death (77) as well as metabolic and obstetrical complications at birth (78,79). As a result, meticulous glycemic control throughout pregnancy is required for optimal maternal and fetal outcomes.
An important first step in achieving optimal glycemic control is to set target BG levels (74,79). However, optimal targets for fasting, preprandial and postprandial BG levels in women with pre-existing diabetes have not been examined in randomized controlled trials; and a variety of BG targets are used in clinical practice. Older studies confirm that the lower the mean BG, the better the outcome, with some suggesting a target mean BG <6.7 mmol/L and, others, a mean <6.9 mmol/L. A fasting BG (FBG) target <5.9 mmol/L is still associated with a 29% macrosomia rate (74,80,81). Recent retrospective data demonstrated that a mean A1C ≥6.0% in pregnant women with type 2 diabetes was associated with increased risk of neonatal complications (preterm birth, neonatal intensive care unit [NICU] admission, neonatal hypoglycemia and jaundice) compared to women with an A1C <6.0% (82). In women with type 1 diabetes and good glycemic control during pregnancy with an A1C of 4.5% to 7.0%, there is still a linear relationship between third trimester A1C and risk of macrosomia (83).
In the absence of comparative studies of specific BG targets for women with pre-existing diabetes, use of the mean BG plus 2 standard deviation (SD) of pregnant women without diabetes appears to be appropriate. This translates into BG targets of fasting and preprandial <5.3 mmol/L; 1 hour postprandial <7.5 mmol/L and 2 hours postprandial <6.7 mmol/L (84). Studies in gestational (GDM) indicate a 1 hour postprandial target <7.8 mmol/L is associated with good pregnancy outcomes (85–89); thus, harmonizing the 1 hour target <7.8 mmol/L is reasonable.
An A1C <6.5% should be strived for in all women with pre-existing diabetes during pregnancy; however, given the slightly increased risk of stillbirth in women with an A1C >6.1% (77), ideally a target A1C ≤6.1% should be sought by the third trimester of pregnancy, if it can be achieved safely.
Definition of hypoglycemia in pregnancy
Hypoglycemia is traditionally defined as a BG <4.0 mmol/L; however, as demonstrated by a group who compared continuous glucose monitoring (CGM) with glucose levels from nonpregnant and pregnant women, BG levels are lower during pregnancy by a factor of 20% (90). By consensus, the American Diabetes Association and Endocrine Society Working Group defined hypoglycemia during pregnancy as a level <3.3 mmol/L (91). However, since the hypoglycemia level is often individualized to each person with diabetes, with consideration of symptoms, therapy, medical condition and associated risk; the official lower limit of BG level during pregnancy is difficult to clearly establish. Overall, it is understood that pregnant women have lower BG values that can be judged as normal even if below the traditional level of 4.0 mmol/L. However, women receiving insulin therapy should maintain BG values >3.7 mmol/L to avoid repeated hypoglycemia.
Hypoglycemia is generally considered to be without risk for the fetus, as demonstrated in women with pre-existing diabetes (79,92–94), as long as it is not sustained and maternal loss of consciousness, convulsion, and fall or trauma is avoided during the episode (91). However, repeated hypoglycemia and associated loss of glycemic control have been associated with macrosomia (95).
The limiting factor when targeting euglycemia in women with pre-existing diabetes is the increased risk of hypoglycemia during pregnancy, particularly in the first trimester (96–100), for both type 1 and type 2 diabetes (79). Up to 71% of pregnant women with pre-existing diabetes may experience severe hypoglycemia, with the major predictors being a history of severe hypoglycemia in the 1-year period preceding pregnancy, diabetes duration >10 years and hypoglycemic unawareness (96–100). The latter may relate, in part, to the loss of counterregulatory hormones reported in women with pre-existing diabetes during pregnancy, particularly growth hormone and epinephrine (95,101–103). This risk of hypoglycemia may be ameliorated if efforts are made to achieve good glycemic control preconception and by the use of analogue insulins (100,104,105) (see Hypoglycemia chapter, p. S104). Health-care providers should ensure that pregnant women with diabetes: a) have a glucagon kit; b) are advised regarding effective interventions if a severe hypoglycemic event occurs; and c) are encouraged to inform close relatives and co-workers of this increased risk, especially in the first and early second trimester.
Monitoring
Frequent self-monitoring of blood glucose (SMBG) in pregnant women with type 1 diabetes is essential during pregnancy in order to achieve the glycemic control associated with better outcomes (80). Preprandial testing (to guide mealtime insulin dose adjustment) and postprandial testing (to meet postprandial targets) are associated with less macrosomia in observational studies and reduced preeclampsia (81,106,107). Due to the increased risk of nocturnal hypoglycemia with any intensive insulin therapy, SMBG during the night is often necessary in pregnant women with diabetes receiving insulin (108). SMBG 4 to 7 times per day is also recommended for pregnant women with type 2 diabetes (i.e. fasting, preprandial and 1 or 2 hours postprandially) to achieve good glycemic control.
CGM may help identify periods of hyper- or hypoglycemia (109,110) and can confirm glycemic variability, especially in women with type 1 diabetes (111). Evidence for the use of CGM to improve glycemic control, and maternal and fetal outcomes is conflicting. One study using blinded, intermittent CGM with review of results with a clinician showed that CGM improved A1C and rates of macrosomia compared to standard care (109). However, a study of intermittent real-time CGM did not demonstrate benefit (112). Finally, a study examining CGM use to prevent episodes of severe hypoglycemia early in pregnancy in women with a history of episodes in the year prior to pregnancy did not demonstrate benefit. The Continuous Glucose Monitoring in Women with Type 1 Diabetes in Pregnancy (CONCEPTT) trial randomized 325 women (215 pregnant and 110 planning pregnancy) to capillary blood glucose (CBG) monitoring with CGM or without. Pregnant CGM users spent more time in target (68% vs. 61%, p=0.0034) and less time hyperglycemic (27% vs. 32%, p=0.0279) than did pregnant control participants, with comparable severe hypoglycemic episodes and time spent hypoglycemic. Neonatal health outcomes were significantly improved, with lower incidence of LGA (OR 0.51, 95% CI 0.28–0.90, p=0.021), fewer NICU admissions lasting more than 24 h (OR 0.48, 95% CI 0.26–0.86, p=0.0157), and fewer incidences of neonatal hypoglycemia (OR 0.45; 95% CI 0.22–0.89, p=0.025. No benefit was observed for women planning a pregnancy (113). Whether closed-loop systems will be beneficial for use in pregnancy remains to be seen (114). One study of pregnant women with type 1 diabetes showed overnight closed-loop therapy resulted in better glycemic control than sensor-augmented pump therapy (115).
Women with pre-existing diabetes during pregnancy should have A1C levels measured during pregnancy to assist in management. A1C levels can also be helpful predictors of adverse pregnancy outcomes (116,117). The optimal frequency of A1C measurement is not known; however, testing more than the usual every 3 months may be appropriate (see Monitoring Glycemic Control chapter, p. S47).
Weight gain
Institute of Medicine (IOM) guidelines for weight gain in pregnancy were first established in 1990 based on neonatal outcomes. Results of a systematic review of studies examining the 1990 IOM recommendations for maternal weight gain in women without diabetes, showed that those who followed guidelines were more likely to have good infant birthweight and fetal growth, and decreased the amount of weight loss required postpartum (118). The IOM revised their recommendations in 2009 due to increasing rates of obesity and to take into consideration maternal obesity; however, IOM recommendations do not take into account pre-existing medical conditions (119).
Cohort studies of various body mass index (BMI) classes of women with pre-existing diabetes showed that excessive gestational weight gain (GWG) is characterized by higher birth weight infants independent of pre-pregnancy BMI and glycemic control (120,121). The researchers suggest that aiming for the lower weight gain range based on BMI category may be useful in the management of women with pre-existing diabetes. Furthermore, prepregnancy overweight and obesity are risk factors for adverse maternal and neonatal outcomes. Findings of cohort studies with pregnant women with type 2 diabetes who had overweight or obesity showed that weight gain greater than the IOM recommendations was associated with increased macrosomia (122–124), LGA (124), adverse neonatal outcomes (123) and higher rates of caesarean deliveries (122,123). The number of women with excessive GWG in these studies ranged from 40% (122) to 70% (124). Studies investigating weight gain below the IOM guidelines in women with obesity and type 2 diabetes have produced conflicting results ranging from: no evidence of worsened perinatal outcomes (122); increased risk of SGA (123); and lower birth weight, LGA and less perinatal morbidity with no increased risk of SGA (125).
Prepregnancy BMI, glycemic control and GWG can have independent and additive effects on fetal growth. Therefore, diabetes education and management for this group of women in preconception and regularly throughout pregnancy should be inclusive of both optimal glycemic control, healthy preconception weight and weight gain through pregnancy. Until additional data on specific weight gain recommendations for women with pre-existing diabetes becomes available, these women should be advised to gain weight as per the IOM guidelines based on their prepregnancy BMI category to lower the risk of LGA, macrosomia and caesarean deliveries.
Pharmacological therapy
Insulin. Insulin therapy must be individualized and regularly adapted to the changing needs of pregnancy (126–129). Intensive insulin therapy with basal-bolus therapy or continuous subcutaneous insulin infusion (CSII or insulin pump) therapy is recommended to achieve glycemic targets prior to pregnancy and during pregnancy. Women using CSII should be educated about the possible increased risk of diabetic ketoacidosis (DKA) in the event of insulin pump failure. However, recent studies using pumps have not demonstrated an increase in DKA compared to multiple daily injections (MDI) (130).
Rapid-acting bolus analogues (e.g. aspart, lispro) appear safe for use in pregnancy, with some studies showing improvement in postprandial glycemia and reduced maternal hypoglycemia compared to regular insulin (131–133). Although there are no studies that have examined placental transfer of aspart, lispro has been examined and does not cross the placenta except at very high doses (>50 units), similar to human insulin (134). A meta-analysis of observational studies (1561 women with pre-existing diabetes and GDM) found that lispro compared to regular insulin was associated with decreased rates of severe maternal hypoglycemia and neonatal jaundice, but increased rates of LGA infants (135). A randomized trial of 322 women with type 1 diabetes randomized to insulin aspart vs. human regular insulin, showed a trend toward reduced episodes of major hypoglycemia, with improved postprandial BG levels but similar overall glycemic control (104). In a smaller, underpowered study, perinatal outcomes were similar using insulin aspart and human insulin (136). A meta-analysis of randomized trials of 1,143 women with gestational or pre-existing diabetes assessing the use of insulin aspart or premixed biphasic insulin aspart 30 compared to human regular insulin or premixed biphasic insulin aspart during pregnancy found similar rates of caesarean section and macrosomia (135). Finally, a case series of 303 women exposed to glulisine during pregnancy showed no noted pattern of few birth defects (137). There are no data to date on faster-acting insulin aspart.
Long-acting insulin analogues, glargine and detemir, appear safe with similar maternal and fetal outcomes compared to neutral protamine hagedorn (NPH) insulin. Both glargine and detemir (138) do not cross the placenta at therapeutic doses, although glargine does cross at very high doses (139). Notably, 2 randomized trials of detemir use compared with NPH in women with type 1 diabetes showed a lowering of FBG, but similar A1C, maternal hypoglycemia, and other maternal and fetal outcomes (140); another trial found less hypoglycemia with detemir compared with NPH (141). The research evidence for glargine are more limited (cohort and case control studies); however, in a meta-analysis of cohort studies comparing glargine to NPH, maternal and fetal outcomes were similar (142) and no adverse maternal or fetal effects have been described to date. Finally, there are no benefit or harms data on the use of glargine insulin U-300, lispro insulin U-200, degludec insulin U-100 and U-200, or glargine biosimilar in pregnancy.
CSII therapy during pregnancy. While the use of CSII may be preferred by some women with type 1 diabetes, older randomized studies have not demonstrated superiority over basal-bolus regimen (132,143–146). A meta-analysis of observational studies comparing the use of CSII (with insulin analogs) to MDI found no differences in maternal or fetal outcomes (147). However, recent studies not included in the meta-analysis suggest improved glycemic control with CSII (148–150), while other studies found no difference (151). Overall, studies show no difference in maternal or fetal outcomes with CSII, but also no increase in harms, such as maternal hypoglycemia, DKA or weight gain. More randomized trials are needed with current CSII technology to better assess the utility of CSII during pregnancy.
Noninsulin antihyperglycemic agents and pregnant women with type 2 diabetes. A meta-analysis of first-trimester use of either glyburide or metformin, and a meta-analysis of metformin alone in women with polycystic ovary syndrome (PCOS) showed no increased incidence of congenital anomalies (152,153). Women with type 2 diabetes who conceive on metformin or glyburide can continue these agents until insulin is initiated. Three smaller randomized trials have examined the use of metformin in pregnant women with type 2 diabetes. The first study was a small, open-label trial in Egypt (n=90) requiring high doses of insulin with poor glycemic control randomized to receive either metformin added to insulin or usual care (154); unfortunately, the authors did not specify whether the women had GDM or type 2 diabetes. The second trial completed in the United States (n=28) involved women with type 2 diabetes randomized to metformin or insulin and showed similar glycemic control in both groups (155). Finally, the third trial completed in Pakistan (n=206) involved women with untreated type 2 diabetes randomized to receive either metformin with insulin (as necessary), or insulin alone (156). In this study, 85% of patients in the metformin group required add-on insulin, but this group experienced less maternal weight gain, less pregnancy-induced hypertension; the infants had an increased rate of small for date, less hypoglycemia and less NICU admissions >24 hours. However, given the small sample sizes in the study and other methodological challenges, the findings from these studies offer limited generalizability.
Currently, a large, double-blind randomized trial is underway to determine whether adding metformin to insulin will benefit mothers with type 2 diabetes and their infants (Metformin in Women with Type 2 Diabetes in Pregnancy [MiTy] and Metformin in Women with Type 2 Diabetes in Pregnancy Kids [MiTy Kids] trials). In conclusion, some studies indicate a possible benefit to adding metformin to insulin in women with type 2 diabetes; however, due to limitations in the research, there is insufficient evidence to recommend the addition of metformin to insulin in pregnant women with type 2 diabetes.
| Table 1 Management of pregnant women with diabetes on insulin receiving betamethasone |
|
|---|---|
| Following the first dose of betamethasone | |
| Day 1 | Increase the night insulin dose by 25% |
| Days 2 and 3 | Increase all insulin doses by 40% |
| Day 4 | Increase all insulin doses by 20% |
| Day 5 | Increase all insulin doses by 10% to 20% |
| Days 6 and 7 | Gradually taper insulin doses to pre-betamethasone doses |
Pregnant women with diabetes receiving steroids. In women suspected of preterm delivery, 2 doses of betamethasone is often given to aid in the maturation of the fetal lungs. The algorithm in Table 1 has been shown to prevent severe hyperglycemia, DKA and severe hypoglycemia in women with type 1 diabetes (157).
Perinatal mortality
Despite health care advances, including NICU, accurate ultrasound dating, SMBG and antenatal steroids for fetal lung maturity, perinatal mortality rates in women with pre-existing diabetes remain increased 1- to 10-fold compared to women without diabetes, and is influenced by glycemic control (1,77). In women with pre-existing diabetes, the risk of stillbirth is higher at all gestations after 32 weeks (158). Perinatal mortality is increased in pregnancies of women with pre-existing diabetes, particularly in those with poor glycemic control (159). In addition, a recent study found that peri-conception A1C >6.6% (adjusted odds ratio [aOR] 1.02), prepregnancy retinopathy (aOR 2.05), lack of prepregnancy folic acid consumption (aOR 2.52) and third-trimester A1C >6.1% (aOR 1.06) were all associated with an increased odds of fetal and infant death (77).
Significance of decreasing insulin requirements
Insulin requirements increase in pregnancy due largely to the “anti-insulin” effects of placental hormones. It has been hypothesized that a marked or rapid decrease in insulin requirements could be a harbinger of placental insufficiency. The relationship between falling insulin requirements and pregnancy outcomes has been explored in 4 retrospective studies (160–163). The 4 studies (n=481 pregnancies) comprised women with type 1 diabetes (n=366), women with type 2 diabetes (n=84), women diagnosed with overt diabetes in pregnancy (n=12) and women with insulin-requiring GDM (n=19). These studies reported decreased insulin requirements (at least 15%) occurred during the third trimester in 8% to 25% of these pregnancies. Only 2 stillbirths occurred: both in the same study of women with pre-existing diabetes (1 in a pregnancy with a >15% decrease in insulin requirements, the other in a pregnancy without a 15% decrease in insulin requirements) (160). This same study found that pregnancies with greater decreases in insulin requirements (>15%) were associated with more SGA neonates and more pre-eclampsia when compared to those that did not have at least a 15% decrease in insulin requirements, suggesting that dropping insulin requirements may be an indicator of placental insufficiency. Those with the ≥15% drop in insulin requirements compared to those without, were delivered slightly earlier at a mean of 37.7 vs. 38.3 gestational weeks. Therefore, not surprisingly, those with the greater decrease in insulin requirements compared to those without, were admitted more frequently to the NICU (23.5% vs. 1.9%, p<0.001). Although care was taken not to include the period within 5 days of antenatal steroid administration when calculating the percent fall in insulin dosing in this study, the substantially higher antenatal steroid use in the pregnancies with falling insulin requirements (31.5% vs. 5.8%, p<0.001) in those without this same fall in insulin requirement suggests that antenatal steroid use may have impacted their retrospective determination of group assignment and, ultimately, their results. However, caution is required in the interpretation of these retrospective studies since decreasing insulin requirements may impact decisions regarding timing of delivery which may, in turn, impact pregnancy outcomes.
In contrast, results from other studies found no association with decreasing insulin requirements and birthweight, and neonatal weight distribution (i.e. SGA to LGA) (161). However, 1 study observed a trend for greater LGA neonates in women with decreasing insulin requirements (162). Caution is required when interpreting the findings as researchers used differing calculation methods to indicate fall in insulin requirements or perhaps due to heterogeneity in the population of women with type 2 diabetes included in the studies. The use of advanced sonographic and fetal doppler assessment in the surveillance of the fetus at risk, as in other high-risk pregnancies, may allow further stratification of risk in this population, but the optimal indicator of feto-placental compromise, particularly in women with diabetes, remains unclear.
In summary, the impact of decreasing insulin requirements is still not certain. While fetal monitoring in this situation can provide reassurance of current fetal well-being, it should not be viewed as a substitute for a well thought out plan for timing of delivery that takes into consideration other risks for perinatal mortality, such as gestational age, maternal glycemic control (both periconception and in later pregnancy), prepregnancy retinopathy (77), maternal age, obesity and smoking history.
Obstetrical considerations in women with pre-existing diabetes and GDM
The goal of fetal surveillance and planned delivery in women with pre-existing diabetes in pregnancy is the reduction of preventable stillbirth. However, not all stillbirths can be avoided due to the fact that many stillbirths in pre-existing diabetes occur prior to 36 weeks of gestation and that in a large number of cases no obvious cause is noted (164). Despite this, it is reasonable to apply surveillance strategies to pre-existing diabetes pregnancies that are similar to those in other pregnancies at high risk of fetal complications, such as intrauterine growth restriction (IUGR), chronic hypertension, and systemic lupus erythematosis (165). Although there is no single strategy for antenatal surveillance for pre-existing diabetes pregnancies, the initiation of some form of fetal surveillance in all women with pregnancies complicated by pre-existing diabetes while applying more intensive protocols for fetal surveillance in pregnancies with additional risk factors is required. These risk factors include: evidence of poor glycemic control, prepregnancy retinopathy (77), LGA, polyhydramnios or the presence of other comorbidities or high-risk conditions (hypertension, obesity, late maternal age, IUGR, previous stillbirth). As a general rule, intensified fetal surveillance should begin at a period in gestation when intervention (i.e. delivery) is possible and acceptable to both the parents and the neonatal care providers.
For GDM, fetal surveillance and timing of delivery are more complex as there is less evidence for increased perinatal mortality in this group. This is likely due to the fact that the risk for perinatal mortality is probably limited to the subgroup of women with poor glycemic control, inclusion of women with pre-existing diabetes in GDM cohorts, obesity and other comorbidities and the rarity of these events. However, a large retrospective cohort (166) showed an increased risk of stillbirth in women with GDM between 36 to 39 weeks of gestation (unadjusted OR 1.1–2.00). Based on the large dataset, a relative risk was calculated of expectant management compared with induction of labour, while taking into consideration both the risk of stillbirth (expectant management) and infant death (expectant management and induction of labour) and showed a significant increased risk of stillbirth with expectant management at both 39 and 40 weeks of gestation when compared with induction of labour. As the absolute risk difference was small, the number needed to deliver to prevent 1 excess perinatal death was estimated as 1,518 at 39 weeks' gestation and 1,311 at 40 weeks' gestation. However, this study is limited by unadjusted confounders, including adequacy and method of glycemic control as well as obesity, thus limiting the generalizability of the study.
There are additional potential benefits of induction of labour in diabetic pregnancies, including reduction of excess fetal growth, shoulder dystocia and caesarean section rate. One randomized controlled trial compared induction of labour with expectant management of labour at term (167). In this trial of insulin requiring GDM and pre-existing diabetes in pregnancies, expectant management after 38 weeks of gestation was associated with increased birthweight and macrosomia, but no change in caesarean section rate. A recent retrospective cohort study from Ontario supports these findings, showing a significant reduction in caesarean section rate at both 38 and 39 weeks of gestation in women with GDM who underwent induction of labour when compared with those that underwent expectant management (168). Conversely, induction of labour at 38 but not 39 weeks was associated with an increase in NICU admission. Importantly, these results remained significant after adjusting for important confounders, including parity, insulin treatment and BMI. Two recently published randomized controlled trials shed additional light on this clinical question. One study randomized women with a suspected macrosomic fetus (>95%) to either induction of labour (IOL) at 37 to 39 weeks or expectant management up until 41 weeks. Although the trial population included diet-controlled GDM (10%), the results showed that IOL resulted in an increased rate of spontaneous vaginal delivery (RR 1.14, 95% Cl 1.01–1.29), a decrease in the rate of shoulder dystocia (RR 0.32 95% Cl 0.12–0.85) and an increase in the rate of neonatal hyperbilirubinemia (169). A second randomized controlled trial randomized women with both diet-controlled and medically treated GDM to IOL at 38 to 38+6 weeks or expectant management until 41 weeks' gestation. The study found no difference in caesarean section rates between groups, but an increase in hyperbilirubinemia was noted in the IOL group. However, the study was underpowered and discontinued due to recruitment difficulties; thus any extrapolations from the study cannot be made (170).
In summary, there is a paucity of quality evidence to guide clinical decisions regarding optimal fetal surveillance and timing of delivery in diabetic pregnancies. Clinical identification of increased risk of stillbirth should be the target of prenatal care and lead to an individualized approach to defining the appropriate regimen of fetal surveillance and timing of delivery. In pre-existing diabetes, poorly controlled GDM or pre-existing diabetes in pregnancy associated with comorbidities, initiation of weekly assessment of fetal well-being at 34 to 36 weeks gestation is recommended. Earlier onset and/or more frequent fetal health surveillance is recommended in those at highest risk. Acceptable methods of assessment of fetal well-being near term can include the nonstress test, amniotic fluid index, biophysical profile or a combination of these. When making decisions regarding timing of delivery before 40 weeks' gestation, the benefits with regards to prevention of stillbirth and a possible decrease in the caesarean rate need to be weighed against the likely increase in neonatal complications.
Glycemic control in labour and delivery
Planning insulin management during labour and delivery is an important part of care and must be adaptable given the unpredictable combination of work of labour, dietary restrictions and need for an operative delivery. The goal is to avoid maternal hypoglycemia while preventing significant hyperglycemia which, in turn, may increase the risk of neonatal hypoglycemia (171). Options for peripartum BG control include watchful waiting until BG rises above a specified threshold (e.g. 7.0 mmol/L for type 2 diabetes or GDM), presumptive initiation of intravenous insulin infusions or continuing with CSII therapy. In a retrospective study of 161 consecutive women with type 1 diabetes, women who chose to continue on CSII during labour had better glycemic control than women using CSII during pregnancy but who chose to convert to intravenous insulin infusion during labour. There was no increase in maternal hypoglycemia, suggesting that the continuation of CSII during labour and delivery appears safe and efficacious (172). Similarly, another retrospective study found that women using CSII had excellent glycemic control without hypoglycemia (173). Observational studies comparing the use of CGM to SMBG during labour and delivery identified improved glycemic control with CGM (173,174); however, neonatal hypoglycemia was comparable between groups (172,174). Each centre should establish protocols which include BG targets, monitoring frequency, insulin regimen and intravenous glucose, based on nursing, medical and anaesthesia expertise available, and patient choice (171,172).
Postpartum care
Postpartum care in women with pre-existing diabetes should include counselling on the following issues: 1) rapid decrease in insulin needs and risk of hypoglycemia in the immediate postpartum period; 2) risk of postpartum thyroid dysfunction in the first months; 3) benefits of breastfeeding; 4) contraceptive measures and; 5) psychosocial assessment and support during this transition period.
Diabetes management and insulin sensitivity immediately postpartum. In women with type 1 and type 2 diabetes, insulin requirements decrease rapidly immediately after the delivery of the placenta (175–177). This rapid increase in insulin sensitivity is related to the drop in circulating placental hormones (hPL, HGH) and, as a result, intravenous insulin infusion or CSII basal insulin should be immediately decreased by at least 50% after delivery to avoid hypoglycemia (175,178).
In the first days postpartum, insulin requirements are generally reduced by an average of 30% to 50% of the prepregnant insulin dosage in women with type 1 diabetes (175–177). In a recent study of 44 women with type 1 diabetes (73% on pumps, 27% on MDI), postpartum total daily insulin was 34% lower than preconception total needs (0.64 to 0.39 units/kg/day postpartum) independent of insulin administration mode or infant feeding. However, a nonsignificant trend toward lower requirements in exclusively breastfeeding mothers compared to partial or full formula feeding was also noted (176). A gradual return to pre-pregnant insulin doses has been noted after 6 to 8 weeks postpartum in some studies (179,180); however, another study found persistently reduced insulin needs up to 4 months postpartum (181). In some studies, reduced insulin needs have been especially noted in women with type 1 diabetes who were breastfeeding (180,181), although this has not been universally observed (176). Nevertheless, most clinicians advise women with type 1 diabetes who are breastfeeding of the potential increased risk of hypoglycemia, especially during night breastfeeding. Thus, for women with pre-existing diabetes in pregnancy, a post-delivery plan for reduced prepregnant insulin dosages, pump settings or noninsulin antihyperglycemic agents should be discussed with the woman and recorded before delivery.
Evidence suggests that despite good glycemic control during pregnancy, continuous weight loss, as well as substantial diabetes education and follow up during pregnancy and in the first months postpartum, glycemic control is managed less effectively by mothers with diabetes in the first year postpartum, and A1C levels gradually increase and return to the pre-pregnancy level (182,183). Postpartum A1C levels are positively associated with pre-pregnancy BMI and postpartum weight retention in type 1 diabetes (182). In addition, most women are unable to return to prepregnancy weight (183). Improved postpartum care and specific interventions for women with pre-existing diabetes should be developed to help women achieve their target weight postpartum (182,183), to improve glycemic control in the first year postpartum (183) and to increase breastfeeding rates (184).
Risk of postpartum thyroid dysfunction. Women with type 1 diabetes are at high risk for autoimmune thyroid disease and, consequently, postpartum thyroid dysfunction. The estimated incidence is as high as 44% among women of childbearing age, and 25% in the first months postpartum (185), representing a 3-fold increase compared to a population without diabetes (185,186). Screening for thyroid hormonal abnormalities during pregnancy and at approximately 3 months postpartum in women with type 1 diabetes is recommended.
Breastfeeding
Lower rate and difficulties around delayed lactation in women with diabetes. A Canadian group demonstrated that women with pre-existing diabetes were less likely to initiate breastfeeding compared with noninsulin-treated mothers with diabetes, GDM women and mothers without diabetes (184). Concordant with other studies (187,188), women with all types of diabetes in pregnancy (GDM, pre-existing, insulin-treated or noninsulin-treated) in this study had also lower rates of exclusive breastfeeding in hospital and on discharge. However, women with pre-existing diabetes were disproportionately affected and had lower rates of breastfeeding (184,189). Lower education and maternal age less than 25 years of age were risk factors associated for lower rates of breastfeeding and exclusive breastfeeding postpartum (184).
Women with pre-existing diabetes tend to have delayed milk production. There is a greater delay in lactation onset in mothers with type 1 diabetes who had poor glycemic control (190). Women with type 1 diabetes also discontinue breastfeeding at a higher rate during the first week postpartum (191–193). Overall, women with any form of diabetes during pregnancy have more nursing difficulties with lower milk supply than women without diabetes (194). However, once established, lactation persists and duration is similar in mothers with and without diabetes (190,195).
There are several pathophysiologic and behavioural explanations for lower breastfeeding rates in women with diabetes. Poor glycemic control, insulin resistance, obesity and impaired bonding between mother and child caused by obstetrical complications (such as NICU admission, prematurity, caesarean section) are the major factors associated with delayed lactation (196). It has been demonstrated that ideal glucose and insulin levels are necessary for lactation (197). Good glycemic control enhances maternal serum and milk prolactin concentrations and decreases the delay in the establishment of lactation that has been observed in mothers with type 1 diabetes (190,198). Maternal obesity has also been correlated with delayed onset of lactogenesis II (>72 hours) postpartum, partly related to the fact that it can alter spontaneous release of prolactin. Moreover, infants of mothers with diabetes showed poorer and immature sucking patterns contributing to the difficulties to breastfeed for those mothers in the first days postpartum (199). Protective factors associated with both higher rates of intention to breastfeed and exclusive breastfeeding included attending antenatal classes and having antenatal care delivered by a health-care provider other than an obstetrician. Indeed, women who received antenatal care from a family physician or other health-care providers were respectively 2 and 3 times more likely to exclusively breastfeed (184). Patient education with prenatal information and postnatal counselling on breastfeeding have been shown to lead to similar breastfeeding rates in women with type 1 diabetes as the population without diabetes (181).
Use of noninsulin antihyperglycemic agents during breastfeeding. Few studies have examined breastfeeding and the use of noninsulin antihyperglycemic agents. Three case series found metformin in the milk and plasma of breastfeeding women who were taking metformin 500 mg 2 or 3 times daily, but infant exposure was well below the 10% “level of concern” (0.182% to 0.65%) (200–202). A study looking at weight, height and motor-social development up to 6 months of age in children of mothers taking metformin while breastfeeding showed normal development and no difference from formula-fed infants (203). One case series that studied women taking glyburide or glipizide while breastfeeding found neither drug in the breastmilk, and the maximum theoretical infant dose was well below 10% (<1.5%), with no hypoglycemia found in the 3 infants tested (204). Although metformin and glyburide can be considered for use during breastfeeding, further long-term studies are needed to better clarify the safety of these drugs. Finally, there are no human studies to date looking at thiazolidinedione (TZD), glucagon-like polypeptide-1 (GLP-1) receptor agonist, dipeptidyl peptidase-4 (DPP-4) inhibitor or sodium-glucose cotransporter-2 (SGLT2) inhibitor use while breastfeeding and, therefore, they should not be taken during breastfeeding.
Use of insulin and newer insulin analogues during breastfeeding. There is no contraindication for women with diabetes treated with insulin to breastfeed (175). Exogenous insulins are excreted into breastmilk, including newer insulin analogues (i.e. aspart, detemir, glargine, glulisine, lispro). Insulin is a normal component of breastmilk (205,206) and similar levels were found in the milk of women with type 1 diabetes, type 2 diabetes and women without diabetes, suggesting an active transport of endogenous and exogenous insulin into breastmilk (207). Insulin normally found in breastmilk of mothers with or without diabetes is thought to be required for intestinal maturation of the infant and could act as a positive modulator of the immune response to insulin as suggested by certain groups (208–210).
Benefits of breastfeeding. Breastfeeding immediately postpartum can be part of an early feeding strategy to reduce the risk of neonatal hypoglycemia in women with diabetes (211). Breastfeeding for more than 4 months has also been shown to be protective against the development of diabetes (OR 0.29, 95% CI 0.13–0.63) at 21 years of age in a cohort of 3,595 young adults (212). It was previously thought that early introduction of cow's milk protein could be involved in the development of beta cell autoimmunity in infants at risk for type 1 diabetes. However, a randomized trial comparing the use of a hydrolyzed formula with smaller foreign proteins, compared with a conventional formula containing cow's milk protein, did not reduce the incidence of diabetes-associated autoantibodies 7 years after exposure in offspring with genetic susceptibility to type 1 diabetes and a family member with type 1 diabetes. These data do not support a short-term benefit from the use of hydrolyzed formula but a longer effect on disease prevalence is under study (213) (see Reducing the Risk of Developing Diabetes chapter, p. S20). Finally, along with other known benefits of breastfeeding for mother and child, although not specific to women with pre-existing diabetes, there is evidence that breastfeeding is a significant protective factor against obesity in children (214–216).
In summary, women with pre-existing diabetes should be encouraged to breastfeed immediately after delivery and for at least 4 months postpartum, as it may contribute to the reduction of neonatal hypoglycemia, offspring obesity and prevent the development of diabetes. Furthermore, exclusive breastfeeding up to 6 months and continuation of breastfeeding up to 2 years with appropriate complementary feeding has shown further benefits and is currently recommended for all women by the Canadian Paediatric Society (217,218). Health-care providers should pay particular attention to promoting breastfeeding in women with diabetes (184,189), especially in the context of maternal obesity, since this high-risk population has the lowest rates of breastfeeding despite demonstrated benefits for mother and child. Attention should be paid, however, to potential increased risk of hypoglycemia, especially during night feeding, in breastfeeding women with type 1 diabetes.
Postpartum contraception
Effective contraception is an important consideration until proper preparation occurs for a subsequent pregnancy in women with pre-existing diabetes. Regarding the choice of a contraceptive method, the same motivations and restrictions apply to women with type 1 and type 2 diabetes as with other women. Evaluation includes discussing women's preferences for a contraceptive method that will ensure compliance. Absolute and relative contraindications to estrogen (breastfeeding, high BP, and microvascular and CV diabetes-related complications) or to an intrauterine device (IUD) (219) also apply. The progesterone-only contraceptive and IUD are safe with breastfeeding (220).
6. GDM
Prevention and risk factors
The incidence of GDM is increasing worldwide. The global prevalence of hyperglycemia during pregnancy has been estimated at 16.9% (21.4 million live births in 2013) using the World Health Organization criteria (221). A higher proportion of women entering pregnancy at an older age and/or with obesity contribute to this increase in prevalence, along with changes in screening strategies and diagnostic criteria. There is a need for an effective and acceptable intervention that will prevent the development of GDM. Such an approach has the potential to improve maternal and child health, with significant savings to the health-care system.
Understanding the pathophysiology of GDM and its risk factors is important for the development of preventive strategies. The GDM population includes a heterogeneous group of women with different metabolic profiles when exposed to pregnancy hormones. Various presentations include:
- Hyperglycemia that likely preceded the pregnancy (e.g. impaired glucose tolerance (IGT), elevated first trimester fasting glucose, overt diabetes in pregnancy, monogenic diabetes)
- Reduced and/or falling insulin secretory capacity (e.g. developing type 1 diabetes)
- Significant insulin resistance from early pregnancy (e.g. polycystic ovary syndrome, women with overweight or obesity, some specific ethnic groups)
- A combination of factors (e.g. family history of diabetes, previous GDM, genetic predisposition for GDM/type 2 diabetes (222,223)).
As insulin sensitivity decreases substantially with pregnancy (224), not all cases of GDM can be prevented. Studies need to focus on identifying the potential groups of women who can benefit from preventive interventions and adapt such strategies to their condition (e.g. preconception vs. during pregnancy, women with obesity or leanness). Considering the heterogeneity of GDM, it seems obvious that tailored recommendations will emerge for each identified group of at-risk women.
More than 30 randomized controlled trials on GDM prevention have been reported. The interventions tested to date include different diets sometimes combined with diverse physical activity plans, vitamin D supplements, myo-inositol, probiotics and metformin. However, only 3 interventions have demonstrated a significant risk reduction for GDM to date. Effective measures included healthy eating, myo-inositol supplementation and probiotic treatment. Among evaluated interventions, diet-based interventions appear to show the most potential for preventing GDM, especially when directed toward women with overweight or obesity as demonstrated in 3 meta-analyses (225–227). The first meta-analysis (225) of 14 randomized controlled trials (n=2,422 pregnant women) compared interventions with standard care in women with risk factors for GDM represented essentially in all studies by maternal overweight and obesity. Interventions evaluated and compared to standard care included diet, physical activity alone, lifestyle changes (diet and physical activity) and metformin. Dietary interventions were associated with a statistically significant lower incidence of GDM (OR 0.33; 95% CI 0.14-0.76) and gestational hypertension (OR 0.28; 95% CI 0.09–0.86) compared to standard care. There was no statistically significant difference in the incidence of GDM or in the secondary outcomes with physical activity alone, lifestyle changes (diet and physical activity) or metformin use compared to standard care. In the 3 randomized controlled trials focusing on diet, a total of 455 women were included, with comparable mean maternal age and mean BMI (36.1 vs. 36.4 kg/m2) in controls. GDM prevalence decreased from 18% to 7% in the diet groups. Healthy eating intervention consisted of a consultation with a trained dietitian, weighing at each antenatal visit and review of food records, but the duration and number of sessions differed among studies. In the second meta-analysis (226), there was a trend toward a reduced risk of GDM in diet-based intervention groups, but a significant reduction in GDM was noted again in subgroup analysis of pregnant women with obesity or overweight (RR 0.40, 95% CI 0.18–0.86). Finally, the composition of protein content of daily meals may be important as a large prospective cohort study demonstrated that an increased prepregnancy intake of animal protein, in particular red meat, was significantly and positively associated with GDM risk, while vegetable protein intake, specifically nuts, was significantly and inversely associated with GDM risk (228).
Mixed-approach interventions, including diet, physical activity and lifestyle modifications, do not appear to prevent GDM in some studies (225,226,229)but seem effective in a recent meta-analysis when introduced before 15 weeks of gestation (227); methodological problems with this study involving the inclusion of studies of diet alone and physical activity alone make this conclusion less reliable and in need of confirmation by further analyses. It can be argued that the complexity of healthy behaviour interventions, the variability of adherence and delay before introduction, as well as the heterogeneity of the maternal metabolic profile and diagnostic criteria in GDM are the main factors that may explain the discrepancies seen and inconclusive evidence for healthy behaviour interventions. Finally, results of meta-analyses on interventions based solely on physical activity programs to prevent GDM are not impressive (small protective effect [230] vs. nonsignificant impact [225]) and studies seem often underpowered with suspected low protocol adherence.
Studies looking at metformin use for GDM reduction in women with obesity (231) and with PCOS (232) have not shown benefit. Moreover, studies are currently insufficient to support clear clinical recommendations regarding vitamin D supplementation in pregnancy to prevent GDM. Only 3 of 8 observational studies (233) and 1 meta-analysis (234)demonstrate a significant inverse relationship between risk of GDM and maternal vitamin D status. Also, incidence of GDM and other obstetrical outcomes were not influenced by vitamin D supplementation (235). Overall, there is currently limited evidence to support lifestyle, physical activity interventions, metformin or vitamin D supplements for GDM prevention.
Probiotics combined with diet and myo-inositol have shown benefit for GDM prevention (226), but these nutritional supplements were assessed in only 1 trial each. One randomized controlled trial demonstrated a 60% GDM reduction with the use of antenatal probiotics, with no impact on GWG (236,237). Moreover, probiotics did not show an impact on glycemic control in GDM women, but attenuated the normal pregnancy-related rise in low-density lipoprotein cholesterol (LDL-C) levels in the third trimester (238). Similar results were obtained with myo-inositol supplements with a 58% risk reduction of developing GDM in pregnant women with overweight or obesity (239,240). However, those studies have been conducted by only 1 research group, with small sample sizes and these results have not been replicated. Before any further recommendations are made for probiotics or myo-inositol supplements for GDM prevention, large randomized trials are needed.
Finally, a recent meta-analysis demonstrated that excessive GWG, occurring in the first and second trimester, increased the risk of GDM by a factor of 1.4, with similar effect in women with normal weight, overweight or obesity (241). Also, BMI increase observed in the inter-pregnancy period in women with normal BMI or with a BMI >27 kg/m2 is associated with higher risk of GDM in their second pregnancy (242). On the other hand, a decrease in inter-pregnancy BMI in women with overweight or obesity significantly decreases their risk of developing GDM in their second pregnancy, reinforcing the importance of a healthy diet and lifestyle during the preconception period for women with overweight or obesity (242). Along these lines, bariatric surgery is becoming increasingly common for the treatment of obesity, and studies looking at pregnancy outcomes following bariatric surgery have found both benefits (decreased GDM, hypertensive disorders, LGA infants) but also some adverse outcomes (SGA infants, preterm deliveries and NICU admissions) (243). As suggested by most experts and the British Obesity and Metabolic Surgery Society (244,245), women should delay pregnancy at least 12 to 18 months after bariatric surgery to limit adverse pregnancy outcomes and allow weight stabilization and replenishing of all vitamins and microelement deficiencies before conception. A study on children born before and after maternal surgical weight loss demonstrated reduced obesity rate and improved cardiometabolic profiles during childhood and adolescence in offspring born after maternal bariatric surgery, positioning bariatric surgery as 1 of the potential options to limit intergenerational transmission of obesity (246).
In summary, evidence is limited but current literature suggests that the only effective GDM preventive measure in early pregnancy that can be considered in high-risk women, especially prepregnant women with obesity, is a healthy diet and close follow up of weight gain to prevent excessive GWG. Nutritional supplements, such as probiotics and myo-inositol, have shown some encouraging results, but these need to be replicated in larger randomized trials. More studies using the same set of diagnostic criteria are needed and focus should be put on specific populations (prepregnant women with obesity, prior GDM and/or PCOS, as well as women with excessive GWG) to be able to develop effective preventive interventions tailored for those high-risk populations to reduce GDM prevalence.
Screening and diagnosis of GDM
Early screening. Screening for diabetes in the first trimester should be considered for diagnosing overt diabetes (diabetes present before pregnancy) in women who are at risk (see Screening for Diabetes chapter, p. S16), including those with a history of previous GDM. The ability to predict abnormal results on glucose screening tests at 24 to 28 weeks and risk of continued dysglycemia postpartum are other, but less compelling, reasons cited to screen in the first trimester.
The test of choice for early screening should be based primarily on the ability to predict poor obstetrical outcomes, which may be modifiable by lifestyle or pharmacological intervention. There are 2 strategies for testing glucose levels in early pregnancy—using the nonpregnancy-recommended screening tests (FPG or A1C) or using the typical 24- to 28-week gestational diabetes screening (50 g glucose challenge test [GCT] and/or 75 g oral glucose tolerance test [OGTT]) criteria (see below). To apply nonpregnant FPG or A1C criteria in early pregnancy does not take into account that both decrease early in pregnancy and may lead to underdiagnosis in women with pre-existing diabetes. On the other hand, there has been no rigorous validation that criteria accepted for the diagnosis of GDM in the second or third trimester are appropriate for use in the first trimester.
First trimester FPG levels are associated with macrosomia and increased caesarean section rates, as well as an increased risk of second-trimester diagnosis of GDM. The results of a retrospective cohort study (n=6,129) suggest that this association between first trimester fasting glucose and later diagnosis of GDM, macrosomia and caesarean section risk is a graded relationship with no clear cut point (247). In another large cohort study (n=17,186 pregnancies), 39% of women with a first trimester FPG over the GDM diagnostic criteria (5.1 mmol/L), will no longer have an elevated FPG if rescreened at 24 to 28 weeks (248). This suggests that first trimester FPG is not reliable for predicting second-trimester GDM.
First-trimester A1C has been used to predict risk of poor obstetrical outcomes, later development of GDM and persistence of postpartum dysglycemia. In 1 study of 16,122 women screened at a median of 47 days gestation, there were higher rates of major congenital anomalies (RR 2.67, 95% Cl 1.28–5.53), preeclampsia (RR 2.42, 95% Cl 1.28–5.53), shoulder dystocia (RR 2.47, 95% CI 1.05–5.85) and perinatal death (RR 3.96,95% Cl 1.54–10.16) with an A1C of 5.9% to 6.4% in the first trimester (249). However, only 23% of women in that study returned for a first-trimester OGTT, highlighting the low uptake of the OGTT in the first trimester. A retrospective cohort study of 2,812 women compared first trimester A1C to 24-week OGTT and found that an A1C of 5.7% to 6.4% had a 13% sensitivity and 94% specificity for predicting GDM based on a second-trimester 75 g OGTT (250). Another recent study in a multiethnic population of 1,156 women who underwent first trimester A1C and 24- to 28-week 2-stage glucose tolerance test, 48 out of 1,180 had an A1C of 5.9% to 6.4%, which was associated with a 3-fold higher rate of preeclampsia (OR 3.539, 95% CI 1.086–11.532) and macrosomia (OR 3.1, 95% CI 1.127–8.603). However, an elevated first trimester A1C shows a low sensitivity (14.5%) but high specificity (97.5%) for predicting second-trimester GDM (251). In a small cohort study of 160 women, the best cut-off for first trimester A1C to differentiate a diagnosis of postpartum type 2 diabetes was ≥5.9% (252). Thus, a first trimester A1C ≥5.9% appears to confer risk of adverse obstetrical outcome, later diagnosis of GDM and postpartum diabetes. Combining a first trimester FPG of 5.1 to 7.0 mmol/L or A1C 5.7% to 6.4%, is more predictive of need for medical management than when GDM is diagnosed later in pregnancy (253).
Although consideration can be given to treatment of women with A1C 5.9% to 6.4% in the first trimester given the evidence of adverse pregnancy outcomes, whether intervention earlier in pregnancy makes a difference remains unknown. In 1 small cohort study, early intervention appeared to lower the risk of preeclampsia (249). A larger cohort trial using a 75 g OGTT for screening high-risk women earlier in pregnancy continued to show higher rates of hypertensive disorders, preterm delivery, caesarean section rates, macrosomia, and neonatal intensive care despite intervention (254). Although widely used before 24 weeks of gestation for assessment of risk in women at high risk of developing GDM, the 75 g OGTT has no validated thresholds for diagnosis of GDM at this gestational age and there is no evidence yet to support a benefit for earlier management in those that screen positive by whatever threshold is used. If an OGTT is performed before 24 weeks of gestation and is negative by the thresholds used to diagnose GDM after 24 weeks, this test needs to be repeated between 24 to 28 weeks.
Finally, all women with diabetes diagnosed during pregnancy, whether diagnosed in the first trimester or later in pregnancy, should be retested postpartum. In 1 study, in women 6 to 8 weeks postpartum who had an A1C ≥6.5% or FPG ≥7.0 at 24 to 28 weeks during pregnancy, 21% had continued diabetes, 37% had impaired fasting glucose (IFG) or IGT and 41% had normal glucose levels (248,250,255).
Screening and diagnosis
As previously outlined in the Canadian Diabetes Association 2013 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada (CPG), Diabetes Canada continues to support universal screening and diagnosis of GDM based on large randomized control trials and meta-analyses demonstrating that treatment of women with GDM reduces fetal overgrowth, shoulder dystocia and preeclampsia (85,256–259). Justification for supporting universal screening for GDM is outlined in detail in the 2013 CPG (260).Assuming universal screening, the method of screening can be either a sequential 2-step or a 1-step process. Methods for sequential screening include the use of glycosuria, A1C, FPG, random plasma glucose (RPG) and a glucose load. Aside from the glucose load, all the other methods mentioned have not been adopted due to their poorer performance as screening tests in most populations (261–267). The most common glucose test used in sequential screening is the 50 g GCT performed between 24 to 28 weeks of gestation, and it is the screening test recommended by Diabetes Canada in the 2013 and 2018 guidelines. The performance of the GCT as a screening test depends on the cut-off values used, the criteria for diagnosis of GDM and the prevalence of GDM in the screened population. As previously discussed in the 2013 CPG, despite its limitations, the 50 g GCT is practical, accepted by pregnant women and caregivers and retains a >98% negative predictive value for GDM in most populations (268). Results from a Canadian prospective study show that sequential screening is associated with lower direct and indirect costs while maintaining equivalent diagnostic power when compared with 1-step testing. Recent observational data demonstrated the feasibility and good uptake of the 2-step approach (269).
An additional question is whether there is a GCT threshold above which GDM can be reliably diagnosed without continuing to the diagnostic OGTT. It is recognized that using a cut-off of ≥11.1 mmol/L after a 50 g GCT will result in a small number of women receiving an erroneous diagnosis of GDM (270). However, these women are at increased risk of adverse perinatal outcomes and might benefit from the same management as those diagnosed with GDM (271), especially since those with a glucose screen >11.1 mmol/L were found to have a 3.7-fold increased rate of insulin treatment compared to women diagnosed as GDM by National Diabetes Data Group (NDDG) or Carpenter and Coustan criteria (272). We thus have decided to maintain the recommendation from the 2013 CPG to diagnose GDM if the glucose level 1 hour after the 50 g GCT is ≥11.1 mmol/L.
What is the optimal method of diagnosis?
Since there is no clear glucose threshold above which pregnancy outcomes responsive to glycemic management occur (268,273,274), controversy persists as to the best diagnostic thresholds to define GDM. The International Association of the Diabetes and Pregnancy Study Groups (IADPSG) Consensus Panel decided to create new diagnostic thresholds for GDM based on data from the Hyperglycemia and Adverse pregnancy Outcome (HAPO) study. IADPSG thresholds are the maternal glucose values from HAPO associated with a 1.75-fold increase of LGA, elevated C-peptide, high neonatal body fat or a combination of these factors, compared with the mean maternal BG values of women studied in HAPO. These arbitrary thresholds, when applied to the HAPO cohort, led to a GDM incidence of 17.8%. The National Institute of Health (NIH) 2013 Consensus Conference summary statement stated that “at present, the panel believes that there is not sufficient evidence to adopt a 1-step approach, such as that proposed by the IADPSG” (275). However, since this publication, national organizations have published guidelines that are divergent in their approach to screening and diagnosis of GDM (276–280), thus perpetuating the international lack of consensus on the criteria for diagnosis of GDM.
Given the lack of agreement that persists in the international community, the 2013 Canadian Diabetes Association Expert Committee acknowledged the controversy and opted to continue to recommend the “preferred” sequential 2-step approach (Figure 1) while recognizing the option of the 1-step IADPSG approach as an “alternative” strategy (Figure 2) (260). The “preferred” approach for sequential screening consists of a 50 g GCT followed by a 75 g OGTT using the glucose thresholds that result in an (also arbitrary) OR of 2.0 for the increased risk of LGA and cord C-peptide (fasting ≥5.3 mmol/L, 1 hour ≥10.6 mmol/L, 2 hours ≥9.0 mmol/L) (273)(Table 2). However, it was recognized that the IADPSG 1-step strategy has the potential to identify a subset of women who would not otherwise be identified as having GDM and could potentially benefit with regards to certain perinatal outcomes. Therefore, a diagnostic strategy consistent with the IADPSG approach of a 1-step 75 g OGTT using the glucose thresholds that result in an OR of 1.75 for the risk of LGA and cord C-peptide was added as an “alternative” method (Figure 2). As outlined in the 2013 CPG, those who believe that all cases of hyperglycemia in pregnancy need to be diagnosed and treated (i.e. increased sensitivity over specificity) will support the use of the 1-step method of GDM diagnosis.
Some data to support Diabetes Canada's “preferred” strategy can be found in an analysis of 1,892 women with mild untreated glucose intolerance (281). In this study, perinatal outcomes for women with 75 g OGTT results that were positive by HAPO 1.75 OR thresholds (Diabetes Canada alternative) were compared to women with 75 g OGTT results that were positive by HAPO 2.0 OR thresholds (Diabetes Canada preferred). LGA rate and birth weight progressively increased with more dysglycemia and were increased in both groups. However, in this study, only women who were positive by HAPO 2.0 OR thresholds had an increased incidence of preeclampsia, preterm delivery, primary caesarean delivery, shoulder dystocia, ponderal index, transient tachypnea and neonatal hypoglycemia after adjustment for confounders (281).
Figure 1
Preferred approach for the screening and diagnosis of gestational diabetes.
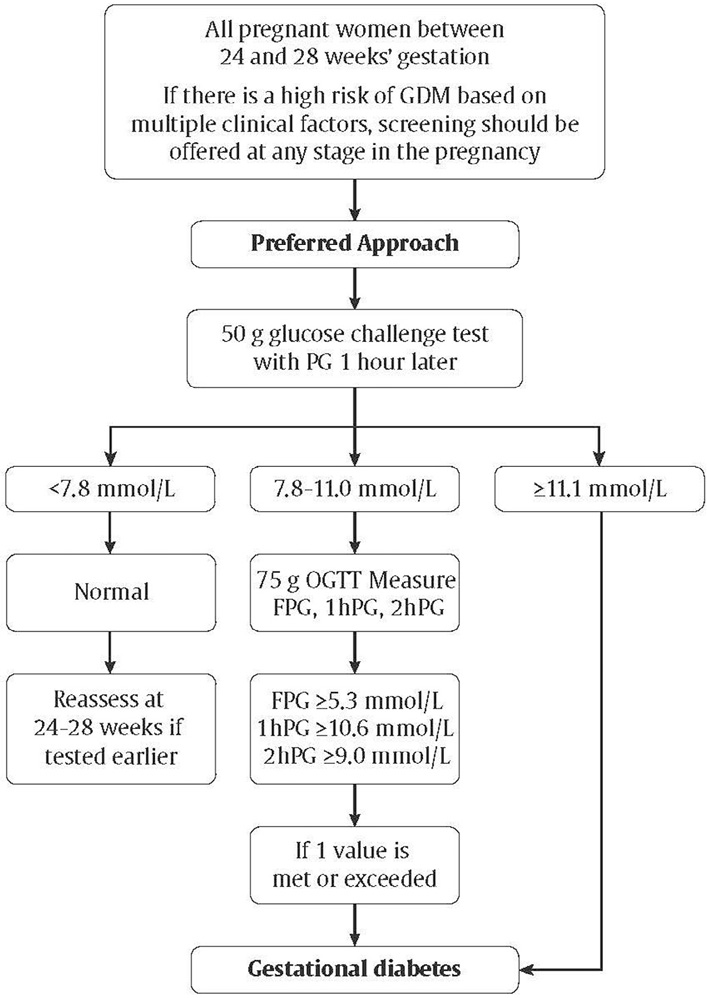
Figure 2
Alternative approach for the screening and diagnosis of gestational diabetes.
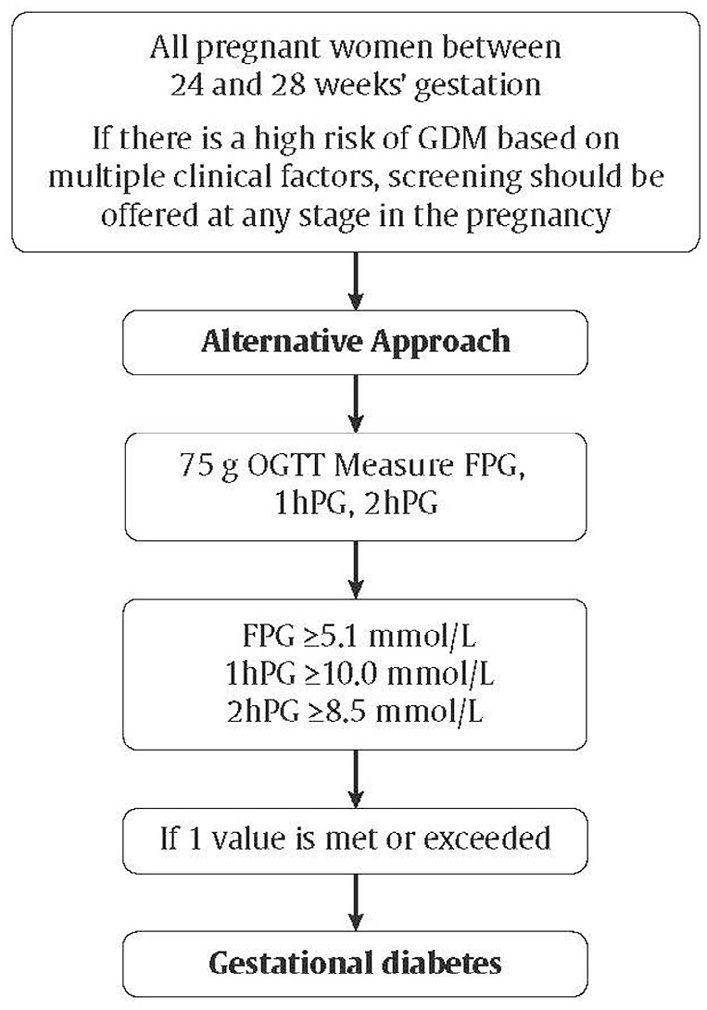
| Table 2 Differences between selecting an OR of 1.75 vs. 2.0 for the primary outcome in the HAPO cohort (273,479) |
||
|---|---|---|
| HAPO, Hyperglycemia and Adverse Pregnancy Outcomes; OR, odds ratio. | ||
| OR 1.75 | OR 2.0 | |
| Threshold glucose levels (mmol/L) | ||
| Fasting | 5.1 | 5.3 |
| 1 hour | 10.0 | 10.6 |
| 2 hour | 8.5 | 9.0 |
| % of HAPO cohort that met ≥1 glucose threshold | 16.1% | 8.8% |
Impact of adoption of IADPSG criteria
Since the publication of the IADPSG consensus thresholds, there have been numerous retrospective studies that have examined the impact of adoption of these criteria. It is difficult to apply the results of these studies to clinical practice due to their retrospective nature and the wide variation in the comparison groups used. In all of these studies, adoption of IADPSG criteria has led to an increase in the number of cases diagnosed while the impact on perinatal outcomes is inconsistent (282–287). Studies comparing pregnancy outcomes before and after changing from a variety of different GDM diagnostic criteria to the IADPSG criteria show differing results. LGA (285) was lower in 1 study and caesarean delivery was lower in several studies (282,285)after adoption of the IADPSG criteria. However, others did not find reductions in LGA (282,283,286,287), and 1 study found an increase in primary caesarean section rate (286).
Given this lack of evidence, it is possible that the decision regarding the recommended screening method will be determined by the economic implications on health-care resources. Decision analysis modelling studies done in other countries (285,288–290) have yielded a variety of results and many are of questionable applicability in the Canadian setting because of differing cost and screening and diagnostic strategies.
A small observational study from Ireland suggested that maternal BMI may be an important consideration in choice of which diagnostic thresholds to use (291). When this group used the IADPSG diagnostic thresholds for all women, they observed a beneficial effect of GDM treatment in women with obesity, but not in women with BMI <25 kg/m2. Furthermore, secondary analysis of the Landon et al trial, that used a 2-step screening approach, found that GDM therapy had a beneficial effect on fetal growth only in women with class 1 and 2 obesity and not in women with normal weight or with more severe obesity (292).
In summary, until more high-quality information comparing the 2013 CPG “preferred” and “alternative” approaches for GDM screening and diagnosis becomes available, the committee agreed it was best to maintain the same diagnostic criteria as those introduced by the 2013 CPG. Further higher-quality evidence would be helpful in establishing if maternal BMI and other clinical risk factors should guide which diagnostic thresholds are used. Most cost analysis evaluations support a sequential screening approach to GDM. The 2018 Diabetes Canada Expert Committee recognizes the drawbacks of having different diagnostic strategies and different thresholds for the same 75 g OGTT but at this time there is insufficient evidence to support 1 strategy over the other (293). Therefore, adequately powered prospective studies to compare these 2 approaches are needed.
Monogenic diabetes in pregnancy
Since pregnancy may be the first time in their lives that women undergo glucose screening, monogenic diabetes may be picked up for the first time in pregnancy. Monogenic diabetes first diagnosed in pregnancy should be suspected in the women with GDM who lack risk factors for GDM and type 1 diabetes and have no autoantibodies (see Definition, Classification, and Diagnosis of Diabetes, Prediabetes and Metabolic Syndrome chapter, p. S10). A detailed family history can be very helpful in determining the likely type of monogenic diabetes. This is important because the type of monogenic diabetes influences fetal risks and management considerations. The most common forms of monogenic diabetes in Canada are maturity onset diabetes of the young (MODY) 2 (heterozygotes for glucokinase [GCK] mutations) or MODY 3 (hepatocyte nuclear factor [HNF] 1 alpha mutation) (294). A history of family members with longstanding isolated elevated FBG with mild A1C elevations that do not progress to “frank” diabetes over a long duration is suggestive of MODY 2. During pregnancy, the usual phenotype for MODY 2 of isolated elevated FBG is not always seen, even though this phenotype may be present outside of pregnancy in the same woman (295). Fetal carriers of GCK mutations (50% of fetuses of an affected parent) do not usually have macrosomia. Fetuses without the GCK mutation of mothers with GCK mutation are at increased risk of macrosomia. The best way to manage women with GCK mutation during pregnancy has yet to be established, but regular fetal growth assessment can aid in the establishment of appropriate glucose targets during pregnancy for women with documented or strongly suspected GCK mutations.
A family history where approximately 50% of family members over 3 generations have diabetes, especially if they are thin and known to be very responsive to insulin secretagogues is highly suggestive of MODY 3 (HNF1 alpha) mutation. MODY 1 (HNF4 alpha mutation) has a similar phenotype to MODY 3 but is much less common. These forms of monogenetic diabetes have greater increased risk of macrosomia and neonatal hypoglycemia that may be prolonged especially in neonates that have MODY 1 (HNF4 alpha mutation). Although women with these later forms of monogenic diabetes are usually exquisitely sensitive to sulfonylureas, they should be transitioned to insulin as they prepare for pregnancy or switched to insulin during pregnancy, if this has not occurred preconception, for the same reasons as avoiding glyburide use in women with GDM.
Management: Healthy behaviour interventions
Weight gain. The 2009 IOM guidelines for weight gain during pregnancy were developed for a healthy population and little is known regarding optimal weight gain in women with GDM. Retrospective cohort studies of GDM pregnancies show that only 31.7% (296) to 42% (297) had GWG within IOM guidelines. Those gaining more than the IOM recommendations had an increased risk of preeclampsia (297), caesarean deliveries (296,297), macrosomia (296,297), LGA (296–298) and GDM requiring pharmacological agents (297). Modification of IOM criteria, including more restrictive targets of weight gain, did not improve perinatal outcomes of interest (296). A large population-based study including women with GDM, concluded that while pre-pregnancy BMI, GDM and excessive GWG are all associated with LGA, preventing excessive GWG has the greatest potential of reducing LGA risk (299). These researchers suggest that, in contrast to obesity and GDM prevention, preventing excessive GWG may be a more viable option as women are closely followed in pregnancy.
A large number of women with overweight or obesity and with GDM gain excessive weight in pregnancy (296,298) and a large proportion exceed their IOM total target by the time of GDM diagnosis (296). A systematic review found that pregnant women with overweight or obesity who gain below the IOM recommendation, but have an appropriately growing fetus, do not have an increased risk of having a SGA infant (118), leading some to recommend that encouraging increased weight gain to conform with IOM guidelines will not improve maternal or fetal outcomes (300). However, other researchers urge caution as they have found that, in women with overweight or obesity and GDM, a weight loss or gain of ≤5 kg was associated with SGA and decreases in neonatal fat mass and lean body mass, including length and head circumference (301). The findings of a retrospective cohort study including women with overweight or obesity and with GDM show that after adjusting for confounding variables, modest weight loss after GDM diagnosis may not adversely impact fetal growth among those in obesity class II/III and those exceeding the IOM guidelines for total GWG at the time of GDM diagnosis (302).
A Cochrane review (49 trials of 11,444 women) was performed to evaluate the effectiveness of diet or exercise or both in preventing excessive gestational weight gain and associated adverse pregnancy outcomes (303). Study interventions involved mainly diet only, exercise only and combined diet and exercise interventions compared with standard care. Results show that diet or exercise or both reduced GWG on average by 20%. Low glycemic load (GL) diets, supervised or unsupervised exercise only or diet and exercise in combination all led to similar reductions in the number of women gaining excessive weight in pregnancy. There was no clear difference between intervention and control groups with regards to preeclampsia, caesarean section, preterm birth and macrosomia. In subgroup analysis by risk, high-risk women (having overweight or obesity, or with or at risk of GDM) who received combined diet and physical activity intervention experienced a 15% reduction in macrosomia.
Further studies are needed to develop weight gain guidelines for GDM patients and to determine whether weight gain less than the IOM guidelines or weight loss in pregnancy is safe. Until this data are available, women with GDM should be encouraged to gain weight as per the IOM guidelines for the BMI category to reduce adverse maternal and neonatal outcomes and postpartum weight retention.
Nutrition therapy. Nutrition therapy is a cornerstone for managing GDM. All women at risk for or diagnosed with GDM should be assessed, counselled and followed up by a registered dietitian when possible (304–306). Nutrition therapy should be designed to promote adequate nutritional intake without ketosis, achievement of glycemic goals, appropriate fetal growth and maternal weight gain (307–310). Recommendations for nutrition best practice (304) and a review of the role of nutrition therapy in GDM management (311) is available.
A great variety of diets are used for managing GDM. While carbohydrate moderation is usually recommended as first-line strategy to achieve euglycemia (312), evidence available to support the use of a low-glycemic-index (GI) diet is increasing. A randomized controlled trial of 70 healthy pregnant women, randomized to low glycemic index (GI) vs. a conventional high-fibre diet from 12 to 16 weeks' gestation, showed a lower prevalence of LGA without an increase in SGA in the low-GI group (313). This led to the hypothesis that a low-GI diet may be beneficial in women with GDM. An earlier systematic review of 9 randomized controlled trials, in which 11 different diet types were assessed within 6 different diet comparisons, did not support the recommendation of 1 diet type over another as no significant differences were noted in macrosomia, LGA or caesarean section rates (314). However, a more recent systematic review and meta-analysis does support the use of low GI diets (315). In this review and meta-analysis of 9 randomized controlled trials (n=884 women with GDM), 3 meta-analyses were performed according to type of dietary intervention used—low-GI, total energy restriction and low-carbohydrate diet. Only the low-GI diet was associated with less frequent insulin use and lower newborn weight without an increase in numbers of SGA and macrosomia (315). Results of a meta-analysis of 5 randomized controlled trials (316) and a systematic review (317) in GDM patients showed that low-GI diets reduce the risk of macrosomia and LGA, respectively. Low-GI diets are associated with lower postprandial blood glucoses in recent randomized controlled trials (318,319).
In summary, current evidence although limited, suggests that women with GDM may benefit from following a low-GI meal pattern (320). Thus, advice on meal planning for women with GDM should emphasize a healthy diet during pregnancy, with a minimum of 175 g/day of carbohydrate (321) distributed over 3 moderate-sized meals and 2 or more snacks (1 of which should be at bedtime), (304,311) as well as replacing high-GI foods with low-GI ones.
Physical activity. In combination with nutritional intervention, physical activity appears to be more effective for GDM management than GDM prevention. A recent review reported that 5 of 7 studies (71%, 5 randomized controlled trials, 1 case-control, 1 self-enrolment) demonstrated a positive impact of physical activity interventions on GDM management by reducing insulin use and/or by improving glycemic control in women with GDM (322). Adherence to the physical activity program was good overall except among the 2 studies that were unsuccessful at improving glycemic control; 1 reported low compliance with physical activity recommendations, and the other proposed an exercise program with a higher level of intensity (>70% of maximal heart rate). No studies had an effect on infant birth weight or macrosomia rate and only 1 was successful in reducing GWG. It can be argued that these studies were not powered enough to demonstrate any impact on birthweight or on adverse pregnancy outcomes. Indeed, relevant limitations for these studies include the following: samples were small (mean of 43 participants per study), participants had different metabolic profiles and risks factors, and different diagnostic criteria for GDM were used.
The best type of intervention that should be recommended is unclear since all the successful programs used different exercise modalities in terms of intensity, type, duration and frequency. More recently, an initiative in India, the Wings Project, demonstrated that an intervention based on increasing total footsteps with pedometers was able to improve glycemic control in 151 women with GDM and reduce adverse neonatal outcomes in the more active tertiles when compared to their GDM counterparts in the upper tertiles of sedentary behaviour (323). Since no exercise-related injuries were experienced during pregnancy in all those studies, physical activity intervention seems safe to recommend.
All together, current knowledge suggests that physical activity interventions in women with GDM should be encouraged unless obstetrical contraindications exist as physical activity may be an important component of GDM management. However, identification of a specific program of physical activity that should be prescribed to GDM women is currently not possible. Further studies are needed involving larger populations to enable the prescription of an evidence-based physical activity intervention.
Glycemic control. In a systematic review of reports of BG levels in non-GDM pregnancies, normal BG levels during later pregnancy (mean and 1 SD above mean) were: fasting 3.9±0.4 mmol/L, 1 hour postprandial 6.1±0.7 mmol/L and 2 hours postprandial 5.5±0.6 mmol/L with a mean BG of 4.9±0.6 mmol/L (84). The peak postprandial BG occurred at 69±24 minutes (84). However, it should be noted that the mean FBG derived from the total of 255 subjects in this report was 0.6 mmol/L lower than that reported in the HAPO study (273). The HAPO study was the largest prospective study of glycemia in pregnancy and reported a mean FBG of 4.5±0.4 mmol/L, derived from 23,316 pregnant women (273). BG levels in pregnant women with obesity without diabetes were slightly higher than their lean counterparts in a study in which CGM was performed in early and late pregnancy after placing pregnant women with obesity or normal weight on a controlled diet (324).
Importantly, it has been demonstrated that the diagnostic OGTT values were not the best predictors of outcomes whereas CBG levels during treatment were strongly correlated to adverse pregnancy outcomes (325). For women with GDM, good outcomes have been reported using targets of FBG <5.3 mmol/L, 1 hour postprandial BG <7.8 mmol/L and 2 hours postprandial <6.7 mmol/L (86–89) and are close to the targets of the 2 randomized controlled trials showing benefit for the treatment of GDM (85,256). Even if BG can normally and physiologically decrease during pregnancy below the traditional level of 4.0 mmol/L, women receiving insulin should maintain BG >3.7 mmol/L to avoid repeated hypoglycemia—see Pre-Existing Diabetes (Type 1 and Type 2) in Pregnancy: Definition of hypoglycemia during pregnancy, for further details. On the other hand, recent studies have questioned the upper limit of the FBG target. A systematic review of 34 studies (9,433 women) suggests that a FBG ≤5.0 mmol/L was associated with a protective effect on the development of macrosomia (OR=0.53, 95% CI 0.31–0.90, p=0.02), LGA (OR=0.68, 95% CI 0.53–0.88, p=0.01), neonatal hypoglycemia (OR 0.65, 95% CI 0.49–0.85, p=0.01), hyperbilirubinemia (OR 0.63, 95% CI 0.43–0.90, p=0.01) and preeclampsia (OR 0.47, 95% CI 0.31–0.72, p=0.01) when it was evaluated for the third trimester (326). Risks of maternal hypoglycemia or fetal low birth weight were not evaluated in this review and adjustment for maternal BMI and different diagnostic criteria for GDM was not performed. However, this meta-analysis supports the work of Rowan et al demonstrating that the lowest risk of complications was seen when fasting CBG was <4.9 mmol/L (mean SD 4.6±0.3 mmol/L) (325). Even if the frequency of SGA infants was lower across the tertile of mean maternal fasting glycemia in this study, SGA rate in women with the lowest mean FBG was not increased and was, in fact, comparable with the rate of the background population. SGA rate was inversely correlated with maternal weight gain before assessment, suggesting that SGA could be partly prevented by adequate follow up of GWG in those women.
Overall, data suggests that a reduced FBG target of ≤5.0 mmol/L for GDM women would limit LGA and other perinatal complications rates. However, large, well-conducted and randomized controlled trials comparing different BG targets are needed to directly address optimal fasting and postprandial BG targets. Further studies should also assess the risk of maternal hypoglycemia, SGA, insulin use and cost-effectiveness of such modification.
Adjustment of glycemic targets based upon fetal abdominal circumference on third-trimester ultrasound
Despite reduced perinatal morbidity with interventions to achieve euglycemia in women with GDM, increased prevalence of macrosomia persists in this population. To improve outcomes, 4 randomized controlled trials (327–329)have examined the use of fetal abdominal circumference (AC) as measured sonographically and regularly in the third trimester to guide medical management of GDM. This approach involves using stricter maternal BG targets (FBG <4.5 and 2-hour postprandial BG <5.6 to 6.1 mmol/L), and an increased use of insulin, if needed, when the fetal AC measures ≥75th percentile (327–329) or ≥70th percentile (330) and conversely relaxed glycemic objectives (FPG <5.6–6.7 and 2-hour postprandial BG <7.8 to 11.1 mmol/L) when risk of LGA was considered low. A recent meta-analysis has shown that this approach can result in a significant 50% reduction in LGA rate (p=0.0017, number needed to treat [NNT] 10 women with GDM) compared to standard care, without an increase in SGA rate (331), but caution should be used before extrapolation of these results to routine clinical practice. Indeed, it may be difficult to apply this flexible approach given the extreme glycemic targets that were used, the fact that routine determination of AC is not done or sufficiently reliable, and frequent ultrasounds may not be accessible to most centres. Further analyses are needed to establish safe stricter and relaxed glycemic targets that should be recommended for women with GDM to limit LGA and SGA rates.
Monitoring
Frequent SMBG is essential to guide therapy of GDM (331,333). Both fasting and postprandial testing are recommended to guide therapy in order to improve fetal outcomes (89,332). CGMS have been useful in determining previously undetected hyperglycemia, but it is not clear if it is cost effective (334–336). Recent randomized controlled trials suggest that CGM may be of benefit in the treatment of GDM. In a randomized trial, 340 women were randomized to undergo blinded 3-day CGM every 2 to 4 weeks from GDM diagnosis at 24 weeks GA or routine care with SMBG (337). Women using CGM had less glucose variability, less BG values out of the target range, as well as less preeclampsia, primary caesarean section and lower infant birthweight.
In a similar study of 106 women with GDM, given CGM from 24 to 28 weeks or 28 weeks to delivery, excess maternal weight gain was reduced in the CGM group compared to women doing only SMBG, especially in women who were treated with CGM earlier, at 24 weeks GA (338). A1C was lower in the CGM group but not statistically significantly different. More studies are needed to assess the benefits of CGM in this population.
In an effort to control their BG by diet, women with GDM may develop starvation ketosis. Older studies raised the possibility that elevated ketoacids may be detrimental to the fetus (94,339). While the clinical significance of these findings are questionable, it appears prudent to avoid ketosis.
eHealth medicine: Telehomecare and new technologies for glucose monitoring and healthy behaviour interventions
Use of new technologies and web-based platforms for BG monitoring in pregnant women with diabetes in Canada and worldwide is rapidly increasing. These initiatives allow for 2-way communication with women monitoring and transmitting their BG results in real time to health-care providers for feedback. Studies have demonstrated 38.0% (340) to 82.7% (341) reduction in face-to-face medical visits and decreased insulin use (340) in pregnant women using telehomecare in conjunction with conventional care, without an increase in maternal or perinatal complications. While 4 studies of GDM women (total n=272) have demonstrated comparable glycemic control and pregnancy outcomes (342–345), other studies with type 1 diabetes (346–348) and GDM (348) have shown improved glycemic control and pregnancy outcomes in the group using web-based programs compared to standard care. Enhanced patient empowerment and greater satisfaction with the care received are also reported in groups using new monitoring technology (340–343,345,348,349). However, generalizability of those studies is questionable as these studies were small, conducted in very specific settings and used different types of technologies and e-platforms. Furthermore, acceptance of these interventions by marginalized population subgroups (350)and in remote regions would also be important to determine. Finally, studies assessing cost effectiveness of these measures, both direct (health system resources utilization) and indirect (work absenteeism, parking, daycare fees) are needed.
Systematic reviews of the literature on the use of technology to support healthy behaviour interventions for healthy pregnant women (351) and women with GDM (352,353)showed that good quality trials in this area are few and research on this topic is in its infancy stage. This is evidenced by the focus on intervention acceptance measures, use of small sample sizes, lack of demonstration of causality and lack of examination of long-term effects or follow up.
In summary, new technologies and telehomecare programs have so far shown encouraging results to reduce medical visits and favour patient empowerment without increasing complication rates in pregnant women with diabetes. In an era of increased prevalence of GDM, well designed and sufficiently powered randomized controlled trials are needed to evaluate the effectiveness of technology as a tool for glucose management, healthy behaviour interventions and a way of relieving health-care system burden.
Pharmacological therapy
Insulin. If women with GDM do not achieve BG targets within 2 weeks of initiation of nutritional therapy and exercise, pharmacological therapy should be initiated (354,355). The use of insulin to achieve glycemic targets has been shown to reduce fetal and maternal morbidity (355,356). A variety of protocols have been used, with multiple daily injections (MDI) being the most effective (357). Insulin usually needs to be continuously adjusted to achieve glycemic targets. Although the rapid-acting bolus analogues aspart and lispro can help achieve postprandial targets without causing severe hypoglycemia (356–358), improvements in fetal outcomes have not been demonstrated with the use of aspart or lispro compared to regular insulin (356,357) (see Pre-Existing Diabetes (Type 1 and Type 2) in Pregnancy: Pharmacological therapy). Glargine and detemir have primarily been assessed in women with pre-existing diabetes in pregnancy (see Pre-Existing Diabetes (Type 1 and Type 2) in Pregnancy: Pharmacological therapy). Randomized trial evidence suggests levemir is safe and may afford less maternal hypoglycemia compared to neutral protamine hagedorn (NPH), while observational studies suggest that glargine, although theoretically less desirable, is also safe.
Other antihyperglycemic agents
Metformin. In several meta-analyses of randomized trials studying the use of metformin compared with insulin in women with gestational diabetes, women treated with metformin had less weight gain (359) and less pregnancy-induced hypertension compared to women treated with insulin (360–365). Infants of mothers using metformin had lower gestational age and less neonatal hypoglycemia. On the other hand, there was conflicting evidence regarding preterm birth, with some studies finding a significant increase with the use of metformin, while others did not. This finding was mainly demonstrated by the Metformin in Gestational diabetes (MiG) trial (366), where there was an increase in spontaneous preterm births rather than iatrogenic preterm births. The reason for this was unclear.
While metformin appears to be a safe alternative to insulin therapy, it does cross the placenta. Results of The Offspring Follow Up of the Metformin in Gestational diabetes (MiG TOFU) trial, at 2 years, showed that the infants exposed to metformin have similar total fat mass but increased subcutaneous fat, suggesting a possible decrease in visceral fat compared to unexposed infants (367). In another follow-up study of infants exposed to metformin during pregnancies with gestational diabetes, children exposed to metformin weighed more at the age of 12 months, and were heavier and taller at 18 months, however, body composition was similar (368) as was motor, social and linguistic development. Studies looking at neurodevelopment showed similar outcomes between exposed and nonexposed infants at 2 years of age (369,370).
In summary, long-term follow up from 18 months to 2 years indicate that metformin exposure in-utero does not seem to be harmful with regards to early motor, linguistic, social, (368), metabolic (367,368) and neurodevelopmental (369,370) outcomes. Longer-term follow up is not yet available.
Glyburide. Glyburide has been shown to cross the placenta. In 2 meta-analyses of randomized trials studying the use of glyburide vs. insulin in women with GDM, glyburide was associated with increased birthweight, macrosomia and neonatal hypoglycemia compared with insulin (361,362). In the same meta-analyses, compared to metformin, glyburide use was associated with increased maternal weight gain, birthweight, macrosomia and neonatal hypoglycemia (361,362). Therefore, the use of glyburide during pregnancy is not recommended as first- or second-line treatment, but may be used as third-line treatment if insulin is declined by the mother and metformin is either declined or insufficient to maintain good glycemic control.
Acarbose. There is only 1 small randomized trial looking at the use of acarbose in women with GDM. There was no difference in maternal/fetal outcomes compared to insulin although gastrointestinal side effects were increased (371).
Other antihyperglycemic agents. There is no human data on the use of DPP-4 inhibitors, GLP-1 receptor agonists or SGLT2 inhibitors. The use of these noninsulin antihyperglycemic agents is not recommended during pregnancy.
7. Obstetrical Considerations in Women with Gestational Diabetes (See Section Entitled ‘Obstetrical Considerations in Women with Pre-Existing Diabetes and Gestational Diabetes’)
Intrapartum glucose management
The primary goal of intrapartum glucose management in women with gestational diabetes is to prevent neonatal hypoglycemia, which is thought to occur from the fetal hyperinsulinism caused by maternal hyperglycemia (372). Longer-term follow-up studies have found that infants with neonatal hypoglycemia had increased rates of neurological abnormalities at 18 months, especially if hypoglycemic seizures occurred or if hypoglycemia was prolonged (373,374) and at 8 years of age with deficits in attention, motor control and perception (375).
Risk of neonatal hypoglycemia is related to maternal BG levels
Maternal hyperglycemia during labour, even when produced for a few hours by intravenous fluids in mothers without diabetes, can cause neonatal hypoglycemia (376,377). Studies have generally been performed in mothers with pregestational diabetes or insulin- treated GDM. These have been observational with no randomized trials deliberately targeting different levels of maternal glycemia during labour. Most have found that there is a continuous relationship between mean maternal BG levels during labour and the risk of neonatal hypoglycemia with no obvious threshold. Authors have often chosen 2 levels within the range and shown that there is more hypoglycemia with the higher value, but the studies do not arrive at a common value and vary from <4.6 mmol/L to <8.0 mmol/L (378–387). By consensus, we suggest aiming for <7.0 mmol/L during labour and delivery.
Intrapartum insulin management
Insulin requirements tend to decrease intrapartum (385,386). There are very few studies (although many published protocols) that examine the best method of managing glycemia during labour (387,388). Given the lack of studies, there are no specific protocols that can be recommended to achieve the desired maternal BG levels during labour.
Postpartum
Breastfeeding. Women with GDM should be encouraged to breastfeed immediately after delivery and for at least 4 months postpartum, as this may contribute to the reduction of neonatal hypoglycemia (211) and offspring obesity (215), and prevent the development of metabolic syndrome and type 2 diabetes in the mother (214,389–397). Longer duration and more intense breastfeeding is associated with less diabetes in the mother with hazard ratios as low as 0.43 (395). Furthermore, offspring that are breastfed for at least 4 months have lower incidence of obesity and diabetes longer term (212). However, GDM is associated with either similar (189) or poor initiation rates (398) compared to those without diabetes, as well as poor continuation rates (189). Factors associated with cessation of breastfeeding before 3 months include breastfeeding challenges at home, return to work, inadequate support, caesarean section and lower socioeconomic status (399). In conclusion, women with GDM should be encouraged to breastfeed as long as possible as intensity and duration of nursing have both infant and maternal benefits (current recommendation by Canadian Paediatric Society is up to 2 years) (217), but more support is needed as this group is at risk for early cessation.
Long-term maternal risk of dysglycemia. With the diagnosis of GDM, there is evidence of impairment of both insulin secretion and action (400,401). These defects persist postpartum and increase the risk of impaired fasting glucose, IGT and type 2 diabetes (402,403). The cumulative risk increases markedly in the first 5 years and more slowly after 10 years (404,405). At 3 to 6 months postpartum, risks of dysglycemia are in the 16% to 20% range. While elevated FPG during pregnancy is a strong predictor of early development of diabetes (406–408), other predictors include age at diagnosis, use of insulin, especially bedtime insulin or oral agents, and more than 2 pregnancies (408–410). A1C at diagnosis of GDM is also a predictor of postpartum diabetes (408,411). Any degree of dysglycemia is associated with increased risk of postpartum diabetes (412). After 16 years, 40% of women with prior GDM will develop type 2 diabetes (413). Some women with GDM, especially lean women under 30 years of age who require insulin during pregnancy, progress to type 1 diabetes (414,415). Women with positive autoantibodies (anti-glutamic acid decarboxylase [anti-GAD], anti-insulinoma antigen 2 [anti- IA2]) are more likely to have diabetes by 6 months postpartum (416).
Postpartum testing is essential to identify women who continue to have diabetes, those who develop diabetes after temporary normalization and those at risk, including those with IGT. However, many women do not receive adequate postpartum follow up, and many believe they are not at high risk for diabetes (417–419). Only 14% to 50% return for postpartum testing (419–422)with annual follow-up rates of only 20% (423,424). Proactive contacts increased testing from 33% to 60% (425,426). Despite this finding, more work in this area is needed to improve uptake. One study revealed that, despite email reminders, absolute improvement was only 10% (427).
Women should be screened postpartum to determine their glucose status. Postnatal FBG has been the most consistently found variable in determining women at high risk for early postpartum diabetes (428). However, FPG alone will miss many women with some degree of abnormal glucose tolerance (429–431); therefore, a 75 g OGTT should be done between 6 weeks and 6 months postpartum. Some recent trials have shown that early postpartum testing (day 2 postpartum) may be as good at detecting diabetes as standard testing times; however, follow up in the standard testing group was poor. One study noted a 100% sensitivity and 94% specificity for diabetes detection but not as effective as identifying other forms of glucose abnormalities, and the sample size was small. If this can be confirmed in more rigorous trials, it may be useful to do early postpartum testing in women at high risk for type 2 diabetes or at high risk for noncompliance with follow up (432). A1C does not have the sensitivity to detect dysglycemia postpartum (433) and, even combined with FBS, did not help improve its sensitivity (434,435).
Women should be counselled that the recurrence rate of GDM is high, from 30% to 84%, in subsequent pregnancies (436,437). Metabolic syndrome has been shown to be more prevalent in women with GDM (438–440) with rates as high as 23%, 3 times age-matched control using IADPSG criteria to diagnose GDM (441). Given the increased risk of CVD (OR 1.51) (442) with metabolic syndrome, consideration should be given to screening for all components of the metabolic syndrome in the postpartum care of women with GDM, especially if there is a family history (443,444). Education on healthy behaviour interventions to prevent diabetes and CVD should begin in pregnancy and continue postpartum (445,446). Awareness of physical activity for prevention of diabetes is low (447), and emphasis on targeted strategies that incorporate women's exercise beliefs may increase participation rates (448). Although 1 study showed women with prior gestational diabetes and IGT reduced their risk of developing diabetes with both a lifestyle intervention or metformin, these women were, on average, 12 years postpartum. More recent intervention studies of women with GDM alone who were closer to the time of delivery were often underpowered and compliance with the intervention was low.
The 2 largest randomized controlled trials to date were conflicting. The Mothers After Gestational Diabetes in Australia (MAGDA) study randomized 573 women within the first year postpartum to a group-based lifestyle intervention vs. standard care. After 1 year they found a 1 kg difference in weight and no difference in waist circumference or FBG (449). However, only 10% of women attended all the sessions, and 34% attended none. In another randomized controlled trial, 260 women were randomized to receive the Mediterranean diet and physical activity sessions for 10 weeks between 3 to 6 months postpartum, and then reinforcement sessions at 9 months, 1, 2 and 3 years. They found that significantly less women developed glycemic disorders in the intervention group (42% vs. 58%) (450). At 3 years, women in the intervention group had a lower BMI and better nutrition but similar rates of physical activity. However, engaging women to adopt health behaviours may be challenging soon after delivery. More studies are needed to explore interventions that may help this population reduce their risk.
Long-term metabolic impact of fetal exposure to maternal GDM.Observational studies have linked maternal GDM with poor metabolic outcomes in offspring (451). However, 3 systematic reviews (452–454) have concluded that maternal GDM is inconsistently or minimally associated with offspring obesity and overweight and this relationship is substantially attenuated or eliminated when adjusted for confounders. The HAPO offspring study extended their follow up to 5- to 7-year-olds and found that after adjustment for maternal BMI, higher maternal plasma glucose (PG) concentrations during pregnancy were not a risk for childhood obesity (455). In contrast, a recent cohort found an association between maternal FPG and offspring BMI at 7 years of age that persisted after adjustment for birth weight, socioeconomic status and maternal pre-pregnancy BMI (456). Current evidence fails to support the hypothesis that treatment of GDM reduces obesity and diabetes in offspring. Three follow-up studies of offspring whose mothers were in randomized controlled trials of GDM management found that treatment of GDM did not affect obesity at 4 to 5 years, 5 to 10 years or a mean age of 9 years (457–459). This follow up may be too short to draw conclusions about longer-term impact. However, it is interesting to note that the excess weight in offspring of women with diabetes in the observational work by Silverman et al (460) was evident by 5 years of age. Furthermore, a subanalysis of another trial follow-up study revealed that comparison by age at follow up 5 to 6 vs. 7 to 10 years old did not influence their findings (458).
Association between maternal diabetes and other long-term offspring outcomes, such as childhood academic achievement and autism spectrum disorders (ASD), have been explored in observational studies. Reassuringly, offspring of mothers with pre-existing type 1 diabetes had similar average grades when finishing primary school compared to matched controls (461). Associations between autism and different types of maternal diabetes during pregnancy have been inconsistent and usually disappear or are substantially attenuated after adjustment for potential confounders (462,463). Unspecified antihyperglycemic medications were either not associated with ASD (463) or not independently associated with ASD risk (462,463), but merit further investigation to assess if there are differences in the association between different types of antihyperglycemic agents and ASD.
Contraception after GDM. Women with prior GDM have numerous choices for contraception. Risk and benefits of each method should be discussed with each patient and same contraindications apply as in non-GDM women. Special attention should be given as women with GDM have higher risk of metabolic syndrome and, if they have risk factors, such as hypertension and other vascular risks, then IUD or progestin-only contraceptives should be considered (464). The effect of progestin-only agents on glucose metabolism and risk of type 2 diabetes in lactating women with prior GDM merits further study as in 1 population this risk was increased (464,465).
Planning future pregnancies. Women with previous GDM should plan future pregnancies in consultation with their health-care providers (466,467). Screening for diabetes should be performed prior to conception to assure normoglycemia at the time of conception (see Screening for Diabetes in Adults chapter, p. S16), and any glucose abnormality should be treated. In an effort to reduce the risk of congenital anomalies and optimize pregnancy outcomes, all women should take a folic acid supplement of 1.0 mg (467).
8. Other Relevant Guidelines
- Screening for Diabetes in Adults, p. S16
- Organization of Diabetes Care, p. S27
- Type 2 Diabetes and Indigenous Peoples, p. S296
Literature Review Flow Diagram for Chapter 36: Diabetes and Pregnancy
*Excluded based on: population, intervention/exposure, comparator/control or study design
From: Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med 6(6): e1000097. doi:10.1371/journal.pmed1000097(480).
For more information, visit www.prisma-statement.org.
9. Author Disclosures
Dr. Feig reports non-financial support from Apotex. Dr. Kader reports personal fees from Eli Lilly, Sanofi, Novo Nordisk, Merck, Janssen, Medtronic, and Hoffman Laroche, outside the submitted work. No other authors have anything to disclose.
Resources
-
Interactive tools
-
PDF
2018 Clinical Practice Guidelines Quick Reference Guide
Includes:Screening @ Diagnosis Quick Reference3 Quick Questions to Ask Patients To Meet Their Goals Quick ReferenceIndividualized Goal Setting...
Recommendations
Pre-existing Diabetes
Preconception care
- All women of reproductive age with type 1 or type 2 diabetes should receive ongoing counselling on reliable birth control, the importance of glycemic control prior to pregnancy, the impact of BMI on pregnancy outcomes, the need for folic acid and the need to stop potentially embryopathic drugs prior to pregnancy [Grade D, Level 4 (7)].
- Women with type 2 diabetes with irregular menses/PCOS who lose significant weight or are started on metformin or a thiazolidinedione (TZD) should be advised that fertility may improve and be counselled regarding possible pregnancy and receive preconception counselling [Grade D, Consensus].
-
Before attempting to become pregnant, women with type 1 or type 2 diabetes should:
- Receive preconception counselling that includes optimal diabetes management, including nutrition, preferably in consultation with an interprofessional pregnancy team to optimize maternal and neonatal outcomes [Grade C, Level 3 (6,7,76,468)]
-
Strive to attain a preconception A1C ≤7.0% (or A1C ≤6.5% if can safely be achieved) to decrease the risk of:
- Spontaneous abortion [Grade C, Level 3 (159)]
- Congenital anomalies [Grade C, Level 3 (7,76,469,470)]
- Preeclampsia [Grade C, Level 3 (471,472)]
- Progression of retinopathy in pregnancy [Grade A, Level 1 for type 1 diabetes (25); Grade D, Consensus for type 2 diabetes]
- Stillbirth [Grade C, Level 3 (77)].
- Supplement their diet with multivitamins containing 1 mg of folic acid at least 3 months preconception and continuing until at least 12 weeks of gestation to prevent congenital anomalies [Grade D, Level 4 (14)]
- Discontinue medications that are potentially embryopathic, including any from the following classes:
- Women on metformin and/or glyburide preconception may continue on these agents if glycemic control is adequate until pregnancy is achieved [Grade C, Level 3 (152,153)]. Women on other antihyperglycemic agents, should switch to insulin prior to conception as there are no safety data for the use of other antihyperglycemic agents in pregnancy [Grade D, Consensus].
Assessment and management of complications
- Women should undergo an ophthalmological evaluation by a vision care specialist during pregnancy planning, the first trimester, as needed during pregnancy after that and, again, within the first year postpartum in order to identify progression of retinopathy [Grade B, Level 1 for type 1 diabetes (25); Grade D, Consensus for type 2 diabetes]. More frequent retinal surveillance during pregnancy as determined by the vision care specialist should be performed for women with more severe pre-existing retinopathy and poor glycemic control, especially those with the greatest anticipatory reductions in A1C during pregnancy, in order to reduce progression of retinopathy [Grade B, Level 1 for type 1 diabetes (25,27); Grade D, Consensus for type 2 diabetes].
- Women with albuminuria or CKD should be followed closely for the development of hypertension and preeclampsia [Grade D, Consensus].
Management in pregnancy
- Once pregnant, women with pre-existing diabetes should receive care by an interprofessional diabetes health-care team, including diabetes educators (nurse and dietitian), obstetrical care provider, and physician/nurse practitioner, with expertise in diabetes and pregnancy to minimize maternal and fetal risks [Grade C, Level 3 (7)].
- Once pregnant, women with type 2 diabetes should be switched to insulin for glycemic control [Grade D, Consensus]. Noninsulin antihyperglycemic agents should only be discontinued once insulin is started [Grade D, Consensus].
-
Pregnant women with pre-existing diabetes should:
- Receive an individualized insulin regimen and glycemic targets typically using intensive insulin therapy by basal-bolus injection therapy [Grade A, Level 1B, for type 1 diabetes (73,129); Grade A, Level 1, (129) for type 2 diabetes] or CSII (insulin pump) [Grade C, Level 3 (147) for type 1 diabetes]
-
Strive for target BG values [Grade D, Consensus for all values]:
- Fasting and preprandial <5.3 mmol/L
- 1 hour postprandial <7.8 mmol/L
- 2 hours postprandial <6.7 mmol/L
- Aim for an A1C of ≤6.5% during pregnancy (≤6.1% if possible), if can be achieved safely, to lower the risk of late stillbirth and infant death [Grade D, Level 4 (77)]
- Be prepared to raise BG and A1C targets in the presence of severe hypoglycemia during pregnancy [Grade D, Consensus]
- Perform SMBG, both pre- and postprandially, to improve pregnancy outcomes [Grade C, Level 3 (76)].
- Health-care providers should discuss appropriate weight gain at the initial visit and regularly throughout pregnancy [Grade D, Consensus]. Recommendations for weight gain during pregnancy should be individualized based on the Institute of Medicine guidelines by pre-pregnancy BMI to lower the risk of LGA infants [Grade B, Level 2 (120,121)].
- Aspart, lispro or glulisine may be used in women with pre-existing diabetes to improve postprandial BG [Grade C, Level 2 (104) for aspart; Grade C, Level 3 (132,133,135) for lispro; Grade D, Level 4 (137) for glulisine] and reduce the risk of severe maternal hypoglycemia [Grade C, Level 3 (135) for aspart and lispro; Grade D, Consensus for glulisine] compared with human regular insulin.
- Detemir [Grade B, Level 2 (474)] or glargine [Grade C, Level 3 (142)] may be used in women with pre-existing diabetes as an alternative to NPH and is associated with similar perinatal outcomes.
- Women with pre-existing diabetes should start ASA 81* mg daily at 12–16 weeks' gestation to reduce the risk of preeclampsia [Grade D, Level 4 (48)]. *81 mg is commonly used in Canada due to its commercial availability, but the optimal dose has yet to be determined. Recent evidence suggests that higher dosage regimens might provide additional efficacy.
- Women with type 1 and insulin-treated type 2 diabetes who receive antenatal corticosteroids to improve fetal lung maturation should follow a protocol that increases insulin doses proactively to prevent hyperglycemia [Grade D, Level 4 (157)] and DKA [Grade D, Consensus].
- Women with type 1 diabetes in pregnancy should be offered use of CGM to improve glycemic control and reduce neonatal complications [Grade B, Level 2 (113)].
Fetal surveillance and timing of delivery
- In women with pre-existing diabetes, assessment of fetal well-being should be initiated at 30–32 weeks' gestation and performed weekly starting at 34–36 weeks' gestation and continued until delivery [Grade D, Consensus]. Earlier onset and/or more frequent fetal health surveillance is recommended in those considered at highest risk [Grade D, Consensus].
- In women with uncomplicated pre-existing diabetes, induction should be considered between 38–39 weeks of gestation to reduce risk of stillbirth [Grade D, Consensus]. Induction prior to 38 weeks of gestation should be considered when other fetal or maternal indications exist, such as poor glycemic control [Grade D, Consensus]. The potential benefit of early term induction needs to be weighed against the potential for increased neonatal complications.
Intrapartum glucose management
- Women should be closely monitored during labour and delivery, and maternal blood glucose levels should be kept between 4.0–7.0 mmol/L in order to minimize the risk of neonatal hypoglycemia [Grade D, Consensus].
- CSII (insulin pump) may be continued in women with pre-existing diabetes during labour and delivery if the women or their partners can independently and safely manage the insulin pump and they choose to stay on the pump during labour and delivery [Grade C, Level 3 (172)for type 1 diabetes; Grade D, Consensus for type 2 diabetes].
Postpartum
- Insulin doses should be decreased immediately after delivery below prepregnant doses and titrated as needed to achieve good glycemic control [Grade D, Consensus].
- Women with pre-existing diabetes should have frequent blood glucose monitoring in the first days postpartum, as they have a high risk of hypoglycemia [Grade D, Consensus].
- For women with pre-existing diabetes, early neonatal feeding should be encouraged immediately postpartum to reduce neonatal hypoglycemia [Grade C, Level 3 (211)]. Breastfeeding should be encouraged to reduce offspring obesity [Grade C, Level 3 (215)] and for a minimum of 4 months to reduce the risk of developing diabetes [Grade C, Level 3 (212)]. Women with pre-existing diabetes should receive assistance and counselling on the benefits of breastfeeding, in order to improve breastfeeding rates, especially in the setting of maternal obesity [Grade D, Consensus ].
- Women with type 1 diabetes should be screened for postpartum thyroiditis with a TSH test at 2–4 months postpartum [Grade D, Consensus].
- Metformin and/or glyburide may be used during breastfeeding [Grade C, Level 3 (203) for metformin; Grade D, Level 4 (204) for glyburide]. Other noninsulin antihyperglycemic agents should not be used during breastfeeding as safety data do not exist for these agents [Grade D, Consensus].
Gestational Diabetes
Prevention
- In women at high risk for GDM based on pre-existing risk factors, nutrition counselling should be provided on healthy eating and prevention of excessive gestational weight gain in early pregnancy, ideally before 15 weeks of gestation, to reduce the risk of developing GDM [Grade B, Level 2 (225,227)].
Screening and Diagnosis
-
Women identified as being at high risk for type 2 diabetes should be offered earlier screening with an A1C test at the first antenatal visit to identify diabetes which may be pre-existing [Grade D, Consensus]. For those women with a hemoglobinopathy or renal disease, the A1C test may not be reliable and screening should be performed with an FPG [Grade D, Consensus]. If the A1C is ≥6.5% or the FPG is ≥7.0 mmol/L, the woman should be considered to have diabetes in pregnancy and the same management recommendations for pre-existing diabetes should be followed [Grade D, Consensus].
- If the initial screening is performed before 24 weeks of gestation and is negative, the woman should be rescreened as outlined in recommendations 28 and 29 between 24–28 weeks of gestation [Grade D, Consensus].
- All pregnant women not known to have pre-existing diabetes should be screened for GDM at 24–28 weeks of gestation [Grade C, Level 3 (475)].
-
The preferred approach for the screening and diagnosis of GDM at 24–28 weeks is the following [Grade D, Consensus]:
- Screening for GDM should be conducted using the 50 g GCT administered in the nonfasting state with PG glucose measured 1 hour later [Grade D, Level 4 (272)]. A PG ≥7.8 mmol/L at 1 hour is a positive screen and is an indication to proceed to the 75 g OGTT [Grade C, Level 2 (268)]. A PG ≥11.1 mmol/L is diagnostic of gestational diabetes and does not require a 75 g OGTT for confirmation [Grade D, Level 4 (272)]
-
If the GCT screen is positive, a 75 g OGTT should be performed as the diagnostic test for GDM using 1 of the following criteria:
- Fasting PG ≥5.3 mmol/L OR
- 1 hour PG ≥10.6 mmol/L OR
- 2 hours PG ≥9.0 mmol/L [Grade B, Level 1 (273)].
-
An alternative approach to screen and diagnose GDM is the 1-step approach: a 75 g OGTT should be performed (with no prior screening 50 g GCT) as the diagnostic test for GDM using 1 of the following criteria:
- Fasting PG ≥5.1 mmol/L OR
- 1 hour PG ≥10.0 mmol/L OR
- 2 hours PG ≥8.5 mmol/L [Grade B, Level 1 (273)].
Management during pregnancy
-
Women with GDM should:
- To improve pregnancy outcomes, strive for target BG values:
- Perform SMBG, both fasting and postprandially, to improve pregnancy outcomes [Grade B, Level 2 (89)]
- For women on insulin therapy, maintain BG levels >3.7 mmol/L [Grade D, Consensus].
- Health-care providers should discuss appropriate weight gain and healthy lifestyle interventions regularly throughout pregnancy [Grade D, Consensus]. Recommendations for weight gain for women with GDM should be individualized based on Institute of Medicine guidelines by pre-pregnancy BMI to prevent excessive gestational weight gain and reduce the risk of LGA [Grade B, Level 2 (297,299)], macrosomia and caesarean sections [Grade B, Level 2 (296,297)].
- Nutritional counselling by a registered dietitian should be provided to women with GDM to help them achieve their nutrition, weight and blood glucose goals [Grade D, Level 4 (306)]. Women with GDM should be encouraged to eat a healthy diet for pregnancy and to replace high-GI foods with low-GI foods to reduce the need for insulin initiation and decrease birth weight [Grade C, Level 3 (315)].
-
If women with GDM do not achieve glycemic targets within 1–2 weeks with nutritional therapy and physical activity, pharmacologic therapy should be initiated [Grade D, Consensus].
- Insulin in the form of basal-bolus injection therapy may be used as first-line therapy [Grade A, Level 1 (129) for insulin]
- Rapid-acting analogue insulin aspart, lispro or glulisine may be used over regular insulin for postprandial glucose control, although perinatal outcomes are similar [Grade B, Level 2 (356,357) for aspart and lispro; Grade D, Consensus for glulisine]
- Metformin may be used as an alternative to insulin [Grade A, Level 1A (362) for metformin]; however, women should be informed that metformin crosses the placenta, longer-term studies are not yet available, and the addition of insulin is necessary in approximately 40% to achieve adequate glycemic control [Grade D, Consensus].
- In women with GDM who decline insulin and do not tolerate or are inadequately controlled on metformin, glyburide may be used [Grade B, Level 2 (362)].
Fetal surveillance and timing of delivery in GDM
- Increased frequency of fetal assessment should be considered in women with GDM that is poorly controlled and/or associated with comorbid conditions [Grade D, Consensus].
- Women with GDM can be offered induction of labour between 38–40 weeks' gestation to potentially reduce the risk of stillbirth [Grade D, Consensus] and the risk of caesarean section [Grade C, Level 2 (167,169)]. Earlier or later induction of labour should be considered based on glycemic control and the presence or absence of other comorbid conditions [Grade D, Consensus].
Intrapartum glucose management
- Women with GDM should be monitored during labour and delivery, and maternal blood glucose levels should be kept between 4.0–7.0 mmol/L in order to minimize the risk of neonatal hypoglycemia [Grade D, Consensus].
Postpartum
- Women with GDM should be encouraged to breastfeed immediately after delivery in order to avoid neonatal hypoglycemia [Grade D, Consensus] and to continue for at least 3–4 months postpartum in order to prevent childhood obesity [Grade C, Level 3 (476)] and diabetes in the offspring [Grade D, Level 4 (476)] and to reduce risk of type 2 diabetes and hypertension in the mother [Grade C, Level 3 (391,395,396,476)].
- Women should be screened with a 75 g OGTT between 6 weeks to 6 months postpartum to detect prediabetes and diabetes [Grade D, Consensus]. Methods to improve postpartum testing, such as phone calls or email reminders to women with a history of GDM, should be employed to improve screening rates [Grade C, Level 3 (425)].
- In women who were diagnosed with diabetes in early pregnancy based on A1C (see recommendation 29), if ongoing hyperglycemia is not evident postpartum, a confirmatory test for diabetes with a FPG or 75 g OGTT should be done at 6 to 8 weeks' postpartum [Grade D, Consensus].
- Women with prior GDM should receive counselling regarding healthy behaviour interventions to reduce the recurrence rate in subsequent pregnancies and reduce their increased risk of type 2 diabetes [Grade C, Level 3 (445,446)].
- In women with prior GDM who have IGT on postpartum screening, healthy behaviour interventions with or without metformin can be used to prevent/delay the onset of diabetes [Grade B, Level 2 (477,478)].
Abbreviations:
A1C, glycated hemoglobin; AC, abdominal circumference; ACE, angiotensin-converting enzyme; aOR; adjusted odds ratio; ARB, angiotensin II receptor blocker; ASD, autism spectrum disorder; BG, blood glucose; BMI, body mass index; BP, blood pressure; CBG; capillary blood glucose; CGM, continuous glucose monitoring; CKD, chronic kidney disease; CrCl; creatinine clearance; CSII; continuous subcutaneous insulin infusion; CV, cardiovascular; CVD, cardiovascular disease; DHC, diabetes health-care; DKA, diabetic ketoacidosis; DPP-4, dipeptidyl peptidase-4; FBG, fasting blood glucose; GCT, glucose challenge test; GDM, gestational diabetes; GLP-1, glucagon-like polypeptide-1; GWG, gestational weight gain; GI, glycemic index; IGT, impaired glucose tolerance; IOL, induction of labour; IOM, Institute of Medicine; IUD, intra-uterine device; LDL-cholesterol, low-density lipoprotein cholesterol; LGA, large for gestational age; MDI, multiple daily injections; MI, myocardial infarct; NICU, neonatal intensive care unit; NNT, number needed to treat; NPH, neutral protamine Hagedorn; OGTT, oral glucose tolerance test; PCOS, polycystic ovarian syndrome; PG, plasma glucose; RAAS; renin angiotensin aldosterone system; RR, relative risk; SD, standard deviation; SGA, small for gestational age; SGLT2, sodium-glucose cotransporter-2; SMBG, self-monitoring of blood glucose; TSH, thyroid-stimulating hormone; TZD, thiazolidinedione.
References
- Feig DS, Hwee J, Shah BR, et al. Trends in incidence of diabetes in pregnancy and serious perinatal outcomes: A large, population-based study in Ontario, Canada, 1996–2010. Diabetes Care 2014;37:1590–6.
- Bell R, Bailey K, Cresswell T, et al. Trends in prevalence and outcomes of pregnancy in women with pre-existing type I and type II diabetes. BJOG 2008;115:445–52.
- CEMACH. Pregnancy in women with type 1 and type 2 diabetes in 2002-03, England, Wales and Northern Ireland. London, UK: Confidential Enquiry into Maternal and Child Health (CEMACH), 2005 http://www.bathdiabetes.org/resources/254.pdf.
- Feig DS, Razzaq A, Sykora K, et al. Trends in deliveries, prenatal care, and obstetrical complications in women with pregestational diabetes: A population-based study in Ontario, Canada, 1996–2001. Diabetes Care 2006;29:232–5.
- Macintosh MC, Fleming KM, Bailey JA, et al. Perinatal mortality and congenital anomalies in babies of women with type 1 or type 2 diabetes in England, Wales, and Northern Ireland: Population based study. BMJ 2006;333:177.
- Wahabi HA, Alzeidan RA, Bawazeer GA, et al. Preconception care for diabetic women for improving maternal and fetal outcomes: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2010;10:63.
- Murphy HR, Roland JM, Skinner TC, et al. Effectiveness of a regional prepregnancy care program in women with type 1 and type 2 diabetes: Benefits beyond glycemic control. Diabetes Care 2010;33:2514–20.
- Lassi ZS, Imam AM, Dean SV, et al. Preconception care: Screening and management of chronic disease and promoting psychological health. Reprod Health 2014;11:S5.
- Owens LA, Egan AM, Carmody L, et al. Ten years of optimizing outcomes for women with type 1 and type 2 diabetes in pregnancy-The Atlantic DIP Experience. J Clin Endocrinol Metab 2016;101:1598–605.
- Evers IM, de Valk HW, Visser GH. Risk of complications of pregnancy in women with type 1 diabetes: Nationwide prospective study in the Netherlands. BMJ 2004;328:915.
- Handisurya A, Bancher-Todesca D, Schober E, et al. Risk factor profile and pregnancy outcome in women with type 1 and type 2 diabetes mellitus. JWomens Health (Larchmt) 2011;20:263–71.
- Persson M, Cnattingius S, Wikstrom AK, et al. Maternal overweight and obesity and risk of pre-eclampsia in women with type 1 diabetes or type 2 diabetes. Diabetologia 2016;59:2099–105.
- Abell SK, Boyle JA, de Courten B, et al. Contemporary type 1 diabetes pregnancy outcomes: Impact of obesity and glycaemic control. Med J Aust 2016;205:162–7.
- Correa A, Gilboa SM, Botto LD, et al. Lack of periconceptional vitamins or supplements that contain folic acid and diabetes mellitus-associated birth defects. Am J Obstet Gynecol 2012;206:218, e1-13.
- Laraia BA, Bodnar LM, Siega-Riz AM. Pregravid body mass index is negatively associated with diet quality during pregnancy. Public Health Nutr 2007;10:920–6.
- Mojtabai R. Body mass index and serum folate in childbearing age women. Eur J Epidemiol 2004;19:1029–36.
- Watkins ML, Rasmussen SA, Honein MA, et al. Maternal obesity and risk for birth defects. Pediatrics 2003;111:1152–8.
- Kachoria R, Oza-Frank R. Receipt of preconception care among women with prepregnancy and gestational diabetes. Diabet Med 2014;31:1690–5.
- Lipscombe LL, McLaughlin HM, Wu W, et al. Pregnancy planning in women with pregestational diabetes. J Matern Fetal Neonatal Med 2011;24:1095–101.
- Endres LK, Sharp LK, Haney E, et al. Health literacy and pregnancy preparedness in pregestational diabetes. Diabetes Care 2004;27:331–4.
- Holing EV, Beyer CS, Brown ZA, et al. Why don’t women with diabetes plan their pregnancies? Diabetes Care 1998;21:889–95.
- Tripathi A, Rankin J, Aarvold J, et al. Preconception counseling in women with diabetes: A population-based study in the north of England. Diabetes Care 2010;33:586–8.
- Kallas-Koeman M, Khandwala F, Donovan LE. Rate of preconception care in women with type 2 diabetes still lags behind that of women with type 1 diabetes. Can J Diabetes 2012;36:170–4.
- Klein BE, Moss SE, Klein R. Effect of pregnancy on progression of diabetic retinopathy. Diabetes Care 1990;13:34–40.
- Diabetes Control and Complications Trial Research Group, The Diabetes Control and Complications Trial Research Group. Effect of pregnancy on microvascular complications in the diabetes control and complications trial. Diabetes Care 2000;23:1084–91.
- Omori Y, Minei S, Testuo T, et al. Current status of pregnancy in diabetic women. A comparison of pregnancy in IDDM and NIDDM mothers. Diabetes Res Clin Pract 1994;24(Suppl.):S273–8.
- Chew EY, Mills JL, Metzger BE, et al. Metabolic control and progression of retinopathy. The Diabetes in Early Pregnancy Study. National Institute of Child Health and Human Development Diabetes in Early Pregnancy Study. Diabetes Care 1995;18:631–7.
- Rasmussen KL, Laugesen CS, Ringholm L, et al. Progression of diabetic retinopathy during pregnancy in women with type 2 diabetes. Diabetologia 2010;53:1076–83.
- Lovestam-Adrian M, Agardh CD, Aberg A, et al. Pre-eclampsia is a potent risk factor for deterioration of retinopathy during pregnancy in Type 1 diabetic patients. Diabet Med 1997;14:1059–65.
- Rosenn B, Miodovnik M, Kranias G, et al. Progression of diabetic retinopathy in pregnancy: Association with hypertension in pregnancy. Am J Obstet Gynecol 1992;166:1214–18.
- Cundy T, Slee F, Gamble G, et al. Hypertensive disorders of pregnancy in women with Type 1 and Type 2 diabetes. Diabet Med 2002;19:482–9.
- Tulek F, Kahraman A, Taskin S, et al. The effects of isolated single umbilical artery on first and second trimester aneuploidy screening test parameters. J Matern Fetal Neonatal Med 2015;28:690–4.
- Egan AM, McVicker L, Heerey A, et al. Diabetic retinopathy in pregnancy: A population-based study of women with pregestational diabetes. J Diabetes Res 2015;2015:7.
- Rahman W, Rahman FZ, Yassin S, et al. Progression of retinopathy during pregnancy in type 1 diabetes mellitus. Clin Exp Ophthalmol 2007;35:231–6.
- Polizzi S, Mahajan VB. Intravitreal anti-VEGF injections in pregnancy: Case series and review of literature. J Ocul Pharmacol Ther 2015;31:605–10.
- Almawi WY, Saldanha FL, Mahmood NA, et al. Relationship between VEGFA polymorphisms and serum VEGF protein levels and recurrent spontaneous miscarriage. Hum Reprod 2013;28:2628–35.
- Galazios G, Papazoglou D, Tsikouras P, et al. Vascular endothelial growth factor gene polymorphisms and pregnancy. J Matern Fetal Neonatal Med 2009;22:371–8.
- Peracha ZH, Rosenfeld PJ. Anti-vascular endothelial growth factor therapy in pregnancy: What we know, what we don’t know, and what we don’t know we don’t know. Retina 2016;36:1413–17.
- Safety labeling changes approved by FDA Center for Drug Evaluation and Research (CDER). Silver Spring: U.S. Food and Drug Administration, 2015 https://www.accessdata.fda.gov/scripts/cder/safetylabelingchanges/. Accessed January2017.
- Feghali M, Khoury JC, Shveiky D, et al. Association of vaginal delivery efforts with retinal disease in women with type I diabetes. J Matern Fetal Neonatal Med 2012;25:27–31.
- Gordin D, Kaaja R, Forsblom C, et al. Pre-eclampsia and pregnancy-induced hypertension are associated with severe diabetic retinopathy in type 1 diabetes later in life. Acta Diabetol 2013;50:781–7.
- Bartsch E, Medcalf KE, Park AL, et al. Clinical risk factors for pre-eclampsia determined in early pregnancy: Systematic review and meta-analysis of large cohort studies. BMJ 2016;353:
- Castiglioni MT, Valsecchi L, Cavoretto P, et al. The risk of preeclampsia beyond the first pregnancy among women with type 1 diabetes parity and preeclampsia in type 1 diabetes. Pregnancy Hypertens 2014;4:34–40.
- Sibai BM, Caritis S, Hauth J, et al. Risks of preeclampsia and adverse neonatal outcomes among women with pregestational diabetes mellitus. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. Am J Obstet Gynecol 2000;182:364–9.
- Schröder W, Heyl W, Hill-Grasshoff B, et al. Clinical value of detecting microalbuminuria as a risk factor for pregnancy-induced hypertension in insulintreated diabetic pregnancies. Eur J Obstet Gynecol Reprod Biol 2000;91:155–8.
- How HY, Sibai B, Lindheimer M, et al. Is early-pregnancy proteinuria associated with an increased rate of preeclampsia in women with pregestational diabetes mellitus? Am J Obstet Gynecol 2004;190:775–8.
- Magee LA, von Dadelszen P, Rey E, et al. Less-tight versus tight control of hypertension in pregnancy. N Engl J Med 2015;372:407–17.
- LeFevre ML, U.S. Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med 2014;161:819–26.
- Tang R, Tang IC, Henry A, et al. Limited evidence for calcium supplementation in preeclampsia prevention: A meta-analysis and systematic review. Hypertens Pregnancy 2015;34:181–203.
- Ekbom P, Damm P, Feldt-Rasmussen B, et al. Pregnancy outcome in type 1 diabetic women with microalbuminuria. Diabetes Care 2001;24:1739–44.
- Dunne FP, Chowdhury TA, Hartland A, et al. Pregnancy outcome in women with insulin-dependent diabetes mellitus complicated by nephropathy. QJM 1999;92:451–4.
- Bagg W, Neale L, Henley P, et al. Long-term maternal outcome after pregnancy in women with diabetic nephropathy. N Z Med J 2003;116:U566.
- Rossing K, Jacobsen P, Hommel E, et al. Pregnancy and progression of diabetic nephropathy. Diabetologia 2002;45:36–41.
- Reece EA, Leguizamon G, Homko C. Stringent controls in diabetic nephropathy associated with optimization of pregnancy outcomes. J Matern Fetal Med 1998;7:213–16.
- Jensen DM, Damm P, Ovesen P, et al. Microalbuminuria, preeclampsia, and preterm delivery in pregnant women with type 1 diabetes: Results from a nationwide Danish study. Diabetes Care 2010;33:90–4.
- Biesenbach G, Grafinger P, Stöger H, et al. How pregnancy influences renal function in nephropathic type 1 diabetic women depends on their pre-conceptional creatinine clearance. J Nephrol 1999;12:41–6.
- Smith MC, Moran P, Ward MK, et al. Assessment of glomerular filtration rate during pregnancy using the MDRD formula. BJOG 2008;115:109–12.
- Koetje PM, Spaan JJ, Kooman JP, et al. Pregnancy reduces the accuracy of the estimated glomerular filtration rate based on Cockroft-Gault and MDRD formulas. Reprod Sci 2011;18:456–62.
- Leguizamon G, Reece EA. Effect of medical therapy on progressive nephropathy: Influence of pregnancy, diabetes and hypertension. J Matern Fetal Med 2000;9:70–8.
- Nielsen LR, Damm P, Mathiesen ER. Improved pregnancy outcome in type 1 diabeticwomen with microalbuminuria or diabetic nephropathy: Effect of intensified antihypertensive therapy? Diabetes Care 2009;32:38–44.
- Purdy LP, Hantsch CE, Molitch ME, et al. Effect of pregnancy on renal function in patients with moderate-to-severe diabetic renal insufficiency. Diabetes Care 1996;19:1067–74.
- Klemetti MM, Laivuori H, Tikkanen M, et al. Obstetric and perinatal outcome in type 1 diabetes patients with diabetic nephropathy during 1988–2011. Diabetologia 2015;58:678–86.
- Nielsen LR, Muller C, Damm P, et al. Reduced prevalence of early preterm delivery in women with Type 1 diabetes and microalbuminuria–possible effect of early antihypertensive treatment during pregnancy. Diabet Med 2006;23:426–31.
- Bogaerts A, Van den Bergh BR, Ameye L, et al. Interpregnancy weight change and risk for adverse perinatal outcome. Obstet Gynecol 2013;122:999–1009.
- Cooper WO, Hernandez-Diaz S, Arbogast PG, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med 2006;354:2443–51.
- Li D-K, Yang C, Andrade S, et al. Maternal exposure to angiotensin converting enzyme inhibitors in the first trimester and risk of malformations in offspring: A retrospective cohort study. BMJ 2011;343:d5931.
- Walfisch A, Al-maawali A, Moretti ME, et al. Teratogenicity of angiotensin converting enzyme inhibitors or receptor blockers. J Obstet Gynaecol 2011;31:465–72.
- Bullo M, Tschumi S, Bucher BS, et al. Pregnancy outcome following exposure to angiotensin-converting enzyme inhibitors or angiotensin receptor antagonists: A systematic review. Hypertension 2012;60:444–50.
- Carrasco M, Rao SC, Bearer CF, et al. Neonatal gabapentin withdrawal syndrome. Pediatr Neurol 2015;53:445–7.
- Guttuso T Jr, Shaman M, Thornburg LL. Potentialmaternal symptomatic benefit of gabapentin and review of its safety in pregnancy. Eur J Obstet Gynecol Reprod Biol 2014;181:280–3.
- Silfen SL, Wapner RJ, Gabbe SG. Maternal outcome in class H diabetes mellitus. Obstet Gynecol 1980;55:749–51.
- Bagg W, Henley PG, Macpherson P, et al. Pregnancy in women with diabetes and ischaemic heart disease. Aust N Z J Obstet Gynaecol 1999;39:99–102.
- The Diabetes Control and Complications Trial Research Group. Pregnancy outcomes in the Diabetes Control and Complications Trial. Am J Obstet Gynecol 1996;174:1343–53.
- Howorka K, Pumprla J, Gabriel M, et al. Normalization of pregnancy outcome in pregestational diabetes through functional insulin treatment and modular out-patient education adapted for pregnancy. Diabet Med 2001;18:965–72.
- Pearson DW, Kernaghan D, Lee R, et al. The relationship between prepregnancy care and early pregnancy loss, major congenital anomaly or perinatal death in type I diabetes mellitus. BJOG 2007;114:104–7.
- Ray JG, O’Brien TE, Chan WS. Preconception care and the risk of congenital anomalies in the offspring of women with diabetes mellitus: A meta-analysis. QJM 2001;94:435–44.
- Tennant PW, Glinianaia SV, Bilous RW, et al. Pre-existing diabetes, maternal glycated haemoglobin, and the risks of fetal and infant death: A populationbased study. Diabetologia 2014;57:285–94.
- Persson M, Norman M, Hanson U. Obstetric and perinatal outcomes in type 1 diabetic pregnancies: A large, population-based study. Diabetes Care 2009;32:2005–9.
- CEMACH. Diabetes in pregnancy: are we providing the best care? Findings of a national enquiry. London: Confidential Enquiry into Maternal and Child Health (CEMACH), 2007. http://www.publichealth.hscni.net/sites/default/files/ Diabetes%20in%20Pregnancy-%20are%20we%20providing%20the%20best%20 care.pdf. Accessed January 2017.
- Langer O, Conway DL. Level of glycemia and perinatal outcome in pregestational diabetes. J Matern Fetal Med 2000;9:35–41.
- Combs CA, Gunderson E, Kitzmiller JL, et al. Relationship of fetal macrosomia to maternal postprandial glucose control during pregnancy. Diabetes Care 1992;15:1251–7.
- Abell SK, Boyle JA, de Courten B, et al. Impact of type 2 diabetes, obesity and glycaemic control on pregnancy outcomes. Aust N Z J Obstet Gynaecol 2017;57:308–14.
- Cyganek K, Skupien J, Katra B, et al. Risk of macrosomia remains glucosedependent in a cohort of women with pregestational type 1 diabetes and good glycemic control. Endocrine 2016;55:447–55.
- Hernandez TL, Friedman JE, Van Pelt RE, et al. Patterns of glycemia in normal pregnancy: Should the current therapeutic targets be challenged? Diabetes Care 2011;34:1660–8.
- Landon MB, Spong CY, Thom E, et al. A multicenter, randomized trial of treatment for mild gestational diabetes. N Engl J Med 2009;361:1339–48.
- Langer O, Berkus M, Brustman L, et al. Rationale for insulin management in gestational diabetes mellitus. Diabetes 1991;40:186–90.
- Langer O, Levy J, Brustman L, et al. Glycemic control in gestational diabetes mellitus–how tight is tight enough: Small for gestational age versus large for gestational age? Am J Obstet Gynecol 1989;161:646–53.
- Rey E, Monier D, Lemonnier MC. Carbohydrate intolerance in pregnancy: Incidence and neonatal outcomes. Clin Invest Med 1996;19:406–15.
- de Veciana M, Major CA, Morgan MA, et al. Postprandial versus preprandial blood glucose monitoring in women with gestational diabetes mellitus requiring insulin therapy. N Engl J Med 1995;333:1237–41.
- Mazze R, Yogev Y, Langer O. Measuring glucose exposure and variability using continuous glucose monitoring in normal and abnormal glucose metabolism in pregnancy. J Matern Fetal Neonatal Med 2012;25:1171–5.
- Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: A report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care 2013;36:1384–95.
- Mills JL, Knopp RH, Simpson JL, et al. Lack of relation of increased malformation rates in infants of diabetic mothers to glycemic control during organogenesis. N Engl J Med 1988;318:671–6.
- Steel JM, Johnstone FD, Hepburn DA, et al. Can prepregnancy care of diabetic women reduce the risk of abnormal babies? BMJ 1990;301:1070–4.
- Churchill JA, Berendes HW, Nemore J. Neuropsychological deficits in children of diabetic mothers. A report from the Collaborative Sdy of Cerebral Palsy. Am J Obstet Gynecol 1969;105:257–68.
- Rosenn BM, Miodovnik M, Khoury JC, et al. Deficient counterregulation: A possible risk factor for excessive fetal growth in IDDM pregnancies. Diabetes Care 1997;20:872–4.
- Rosenn BM, Miodovnik M, Holcberg G, et al. Hypoglycemia: The price of intensive insulin therapy for pregnant women with insulin-dependent diabetes mellitus. Obstet Gynecol 1995;85:417–22.
- Evers IM, ter Braak EW, de Valk HW, et al. Risk indicators predictive for severe hypoglycemia during the first trimester of type 1 diabetic pregnancy. Diabetes Care 2002;25:554–9.
- Nielsen LR, Pedersen-Bjergaard U, Thorsteinsson B, et al. Hypoglycemia in pregnant women with type 1 diabetes: Predictors and role of metabolic control. Diabetes Care 2008;31:9–14.
- Robertson H, Pearson DW, Gold AE. Severe hypoglycaemia during pregnancy in women with Type 1 diabetes is common and planning pregnancy does not decrease the risk. Diabet Med 2009;26:824–6.
- Heller S, Damm P, Mersebach H, et al. Hypoglycemia in type 1 diabetic pregnancy: Role of preconception insulin aspart treatment in a randomized study. Diabetes Care 2010;33:473–7.
- Diamond MP, Reece EA, Caprio S, et al. Impairment of counterregulatory hormone responses to hypoglycemia in pregnant women with insulindependent diabetes mellitus. Am J Obstet Gynecol 1992;166:70–7.
- Rosenn BM, Miodovnik M, Khoury JC, et al. Counterregulatory hormonal responses to hypoglycemia during pregnancy. Obstet Gynecol 1996;87:568–74.
- Björklund A, Adamson U, Andreasson K, et al. Hormonal counterregulation and subjective symptoms during induced hypoglycemia in insulin-dependent diabetes mellitus patients during and after pregnancy. Acta Obstet Gynecol Scand 1998;77:625–34.
- Mathiesen ER, Kinsley B, Amiel SA, et al. Maternal glycemic control and hypoglycemia in type 1 diabetic pregnancy: A randomized trial of insulin aspart versus human insulin in 322 pregnant women. Diabetes Care 2007;30:771–6.
- Negrato CA, Rafacho A, Negrato G, et al. Glargine vs. NPH insulin therapy in pregnancies complicated by diabetes: An observational cohort study. Diabetes Res Clin Pract 2010;89:46–51.
- Manderson JG, Patterson CC, Hadden DR, et al. Preprandial versus postprandial blood glucose monitoring in type 1 diabetic pregnancy: A randomized controlled clinical trial. Am J Obstet Gynecol 2003;189:507–12.
- Jovanovic-Peterson L, Peterson CM, Reed GF, et al. Maternal postprandial glucose levels and infant birth weight: The Diabetes in Early Pregnancy Study. The National Institute of Child Health and Human Development–Diabetes in Early Pregnancy Study. Am J Obstet Gynecol 1991;164:103–11.
- Egger M, Davey Smith G, Stettler C, et al. Risk of adverse effects of intensified treatment in insulin-dependent diabetes mellitus: Ameta-analysis. Diabet Med 1997;14:919–28.
- Murphy HR, Rayman G, Lewis K, et al. Effectiveness of continuous glucose monitoring in pregnant women with diabetes: Randomised clinical trial. BMJ 2008;337:a1680.
- Murphy HR, Rayman G, Duffield K, et al. Changes in the glycemic profiles of women with type 1 and type 2 diabetes during pregnancy. Diabetes Care 2007;30:2785–91.
- Kerssen A, de Valk HW, Visser GH. Day-to-day glucose variability during pregnancy in women with Type 1 diabetes mellitus: Glucose profiles measured with the Continuous Glucose Monitoring System. BJOG 2004;111:919–24.
- Secher AL, Ringholm L, Andersen HU, et al. The effect of real-time continuous glucose monitoring in pregnant women with diabetes: A randomized controlled trial. Diabetes Care 2013;36:1877–83.
- Feig DS, Donovan LE, Corcoy R, et al. Continuous glucose monitoring in pregnant women with type 1 diabetes (CONCEPTT): A multicentre international randomised controlled trial. Lancet 2017;Ahead of Print.
- Murphy HR, Elleri D, Allen JM, et al. Closed-loop insulin delivery during pregnancy complicated by type 1 diabetes. Diabetes Care 2011;34:406–11.
- Stewart ZA, Wilinska ME, Hartnell S, et al. Closed-Loop insulin delivery during pregnancy in women with type 1 diabetes. N Engl J Med 2016;375:644–54.
- Fox NS, Roman AS, Saltzman DH, et al. Obesity and adverse pregnancy outcomes in twin pregnancies. J Matern Fetal Neonatal Med 2014;27:355–9.
- Maresh MJ, Holmes VA, Patterson CC, et al. Glycemic targets in the second and third trimester of pregnancy for women with type 1 diabetes. Diabetes Care 2015;38:34–42.
- Siega-Riz AM, Viswanathan M, Moos MK, et al. A systematic review of outcomes of maternal weight gain according to the Institute of Medicine recommendations: Birthweight, fetal growth, and postpartum weight retention. Am J Obstet Gynecol 2009;201:339, e1-14.
- Institute of Medicine (US) and National Research Council (US) Committee to Reexamine IOM Pregnancy Weight Guidelines. Weight gain during pregnancy: reexamining the guidelines. Washington, DC: (US) NAP, 2009
- Secher AL, Parellada CB, Ringholm L, et al. Higher gestational weight gain is associated with increasing offspring birth weight independent of maternal glycemic control in women with type 1 diabetes. Diabetes Care 2014;37:2677–84.
- Parellada CB, Asbjornsdottir B, Ringholm L, et al. Fetal growth in relation to gestational weight gain in women with type 2 diabetes: An observational study. Diabet Med 2014;31:1681–9.
- Yee LM, Cheng YW, Inturrisi M, et al. Effect of gestational weight gain on perinatal outcomes in women with type 2 diabetes mellitus using the 2009 Institute of Medicine guidelines. Am J Obstet Gynecol 2011;205:257, e1-6.
- Harper LM, Shanks AL, Odibo AO, et al. Gestational weight gain in insulinresistant pregnancies. J Perinatol 2013;33:929–33.
- Siegel AM, Tita A, Biggio JR, et al. Evaluating gestational weight gain recommendations in pregestational diabetes. Am J Obstet Gynecol 2015;213:563, e1-5.
- Asbjörnsdóttir B, Rasmussen SS, Kelstrup L, et al. Impact of restricted maternal weight gain on fetal growth and perinatal morbidity in obese women with type 2 diabetes. Diabetes Care 2013;36:1102–6.
- National Collaborating Centre forWomen’s and Children’s Health, National Institute for Health and Clinical Excellence (NICE). Diabetes in pregnancy. Management of diabetes and Its complications from pre-conception to the postnatal period. London: RCOG Press at the Royal College of Obstetricians and Gynaecologists, 2008. https://www.nice.org.uk/guidance/cg63/evidence. Accessed January 2017.
- Quevedo SF, Coustan DR. Diabetes and pregnancy. Use of an integrated “team” approach provides the necessary comprehensive care. R I Med J 1989;72:129–32.
- Jovanovic L, Druzin M, Peterson CM. Effect of euglycemia on the outcome of pregnancy in insulin-dependent diabetic women as compared with normal control subjects. Am J Med 1981;71:921–7.
- Nachum Z, Ben-Shlomo I, Weiner E, et al. Twice daily versus four times daily insulin dose regimens for diabetes in pregnancy: Randomised controlled trial. BMJ 1999;319:1223–7.
- Chauhan SP, Perry KG Jr, McLaughlin BN, et al. Diabetic ketoacidosis complicating pregnancy. J Perinatol 1996;16:173–5.
- Banerjee M, Bhattacharya A, Hughes SM, et al. Efficacy of insulin lispro in pregnancies complicated with pregestational diabetes mellitus. Pract Diabetes Int 2009;26:366–70.
- Chico A, Saigi I, Garcia-Patterson A, et al. Glycemic control and perinatal outcomes of pregnancies complicated by type 1 diabetes: Influence of continuous subcutaneous insulin infusion and lispro insulin. Diabetes Technol Ther 2010;12:937–45.
- Durnwald CP, Landon MB. A comparison of lispro and regular insulin for the management of type 1 and type 2 diabetes in pregnancy. J Matern Fetal Neonatal Med 2008;21:309–13.
- Boskovic R, Feig DS, Derewlany L, et al. Transfer of insulin lispro across the human placenta: In vitro perfusion studies. Diabetes Care 2003;26:1390–4.
- Lv S, Wang J, Xu Y. Safety of insulin analogs during pregnancy: A metaanalysis. Arch Gynecol Obstet 2015;292:749–56.
- Hod M, Damm P, Kaaja R, et al. Fetal and perinatal outcomes in type 1 diabetes pregnancy: A randomized study comparing insulin aspart with human insulin in 322 subjects. Am J Obstet Gynecol 2008;198:186, e1-7.
- Doder Z, Vanechanos D, Oster M, et al. Insulin glulisine in pregnancy—experience from clinical trials and post-marketing surveillance. Eur Endocrinol 2015;11:17–20.
- Suffecool K, Rosenn B, Niederkofler EE, et al. Insulin detemir does not cross the human placenta. Diabetes Care 2015;38:e20–1.
- McCance DR, Damm P, Mathiesen ER, et al. Evaluation of insulin antibodies and placental transfer of insulin aspart in pregnant women with type 1 diabetes mellitus. Diabetologia 2008;51:2141–3.
- Mathiesen ER, Hod M, Ivanisevic M, et al. Maternal efficacy and safety outcomes in a randomized, controlled trial comparing insulin detemir with NPH insulin in 310 pregnant women with type 1 diabetes. Diabetes Care 2012;35:2012–17.
- Herrera KM, Rosenn BM, Foroutan J, et al. Randomized controlled trial of insulin detemir versus NPH for the treatment of pregnant women with diabetes. Am J Obstet Gynecol 2015;213:426, e1-7.
- Pollex E, Moretti ME, Koren G, et al. Safety of insulin glargine use in pregnancy: A systematic review and meta-analysis. Ann Pharmacother 2011;45:9–16.
- Cohen O, Keidar N, Simchen M, et al. Macrosomia in well controlled CSII treated Type I diabetic pregnancy. Gynecol Endocrinol 2008;24:611–13.
- Farrar D, Tuffnell DJ,West J. Continuous subcutaneous insulin infusion versus multiple daily injections of insulin for pregnant women with diabetes. Cochrane Database Syst Rev 2007;(6):CD005542.
- Kernaghan D, Farrell T, Hammond P, et al. Fetal growth in women managed with insulin pump therapy compared to conventional insulin. Eur J Obstet Gynecol Reprod Biol 2008;137:47–9.
- Chen R, Ben-Haroush A,Weismann-Brenner A, et al. Level of glycemic control and pregnancy outcome in type 1 diabetes: A comparison between multiple daily insulin injections and continuous subcutaneous insulin infusions. Am J Obstet Gynecol 2007;197:404, e1-5.
- Ranasinghe PD, Maruthur NM, Nicholson WK, et al. Comparative effectiveness of continuous subcutaneous insulin infusion using insulin analogs and multiple daily injections in pregnant women with diabetes mellitus: A systematic review and meta-analysis. J Womens Health 2015;24:237–49.
- Kallas-Koeman MM, Kong JM, Klinke JA, et al. Insulin pump use in pregnancy is associated with lower HbA1c without increasing the rate of severe hypoglycaemia or diabetic ketoacidosis in women with type 1 diabetes. Diabetologia 2014;57:681–9.
- Neff KJ, Forde R, Gavin C, et al. Pre-pregnancy care and pregnancy outcomes in type 1 diabetes mellitus: A comparison of continuous subcutaneous insulin infusion and multiple daily injection therapy. Ir J Med Sci 2014;183:397–403.
- Kekäläine P, Juuti M, Walle T, et al. Continuous subcutaneous insulin infusion during pregnancy in women with complicated type 1 diabetes Is associated with better glycemic control but not with improvement in pregnancy outcomes. Diabetes Technol Ther 2016;18:144–50.
- Mello G, Biagioni S, Ottanelli S, et al. Continuous subcutaneous insulin infusion (CSII) versus multiple daily injections (MDI) of rapid-acting insulin analogues and detemir in type 1 diabetic (T1D) pregnant women. J Matern Fetal Neonatal Med 2015;28:276–80.
- Gutzin SJ, Kozer E, Magee LA, et al. The safety of oral hypoglycemic agents in the first trimester of pregnancy: A meta-analysis. Can J Clin Pharmacol 2003;10:179–83.
- Cassina M, Dona M, Di Gianantonio E, et al. First-trimester exposure to metformin and risk of birth defects: A systematic review and meta-analysis. Hum Reprod Update 2014;20:656–69.
- Ibrahim MI, Hamdy A, Shafik A, et al. The role of adding metformin in insulin-resistant diabetic pregnant women: A randomized controlled trial. Arch Gynecol Obstet 2014;289:959–65.
- Hickman MA, McBride R, Boggess KA, et al. Metformin compared with insulin in the treatment of pregnant women with overt diabetes: A randomized controlled trial. Am J Perinatol 2013;30:483–90.
- Ainuddin JA, Karim N, Zaheer S, et al. Metformin treatment in type 2 diabetes in pregnancy: An active controlled, parallel-group, randomized, open label study in patients with type 2 diabetes in pregnancy. J Diabetes Res 2015;2015:325851.
- Mathiesen ER, Christensen AB, Hellmuth E, et al. Insulin dose during glucocorticoid treatment for fetal lung maturation in diabetic pregnancy: Test of an algorithm [correction of analgoritm]. Acta Obstet Gynecol Scand 2002;81:835–9.
- Holman N, Bell R, Murphy H, et al. Women with pre-gestational diabetes have a higher risk of stillbirth at all gestations after 32 weeks. Diabet Med 2014;31:1129–32.
- Inkster ME, Fahey TP, Donnan PT, et al. Poor glycated haemoglobin control and adverse pregnancy outcomes in type 1 and type 2 diabetes mellitus: Systematic review of observational studies. BMC Pregnancy Childbirth 2006;6:30.
- Padmanabhan S, McLean M, Cheung NW. Falling insulin requirements are associated with adverse obstetric outcomes in women with preexisting diabetes. Diabetes Care 2014;37:2685–92.
- Achong N, Callaway L, d’Emden M, et al. Insulin requirements in late pregnancy in women with type 1 diabetes mellitus: A retrospective review. Diabetes Res Clin Pract 2012;98:414–21
- McManus RM, Ryan EA. Insulin requirements in insulin-dependent and insulinrequiring GDM women during final month of pregnancy. Diabetes Care 1992;15:1323–7.
- Steel JM, Johnstone FD, Hume R, et al. Insulin requirements during pregnancy in women with type I diabetes. Obstet Gynecol 1994;83:253–8.
- Lauenborg J, Mathiesen E, Ovesen P, et al. Audit on stillbirths in women with pregestational type 1 diabetes. Diabetes Care 2003;26:1385–9.
- Alfirevic Z, Stampalija T, Gyte GM. Fetal and umbilical Doppler ultrasound in high-risk pregnancies. Cochrane Database Syst Rev 2010;(1):CD007529.
- Rosenstein MG, Cheng YW, Snowden JM, et al. The risk of stillbirth and infant death stratified by gestational age in women with gestational diabetes. Am J Obstet Gynecol 2012;206:309, e1-7.
- Kjos SL, Henry OA, Montoro M, et al. Insulin-requiring diabetes in pregnancy: A randomized trial of active induction of labor and expectant management. Am J Obstet Gynecol 1993;169:611–15.
- Melamed N, Ray JG, Geary M, et al. Induction of labor before 40 weeks is associated with lower rate of cesarean delivery in women with gestational diabetes mellitus. Am J Obstet Gynecol 2016;214:364, e1-8.
- Boulvain M, Senat MV, Perrotin F, et al. Induction of labour versus expectant management for large-for-date fetuses: A randomised controlled trial. Lancet 2015;385:2600–5.
- Alberico S, Erenbourg A, Hod M, et al. Immediate delivery or expectant management in gestational diabetes at term: The GINEXMAL randomised controlled trial. BJOG 2017;124:669–77.
- Ryan EA, Al-Agha R. Glucose control during labor and delivery. Curr Diab Rep 2014;14:450.
- Drever E, Tomlinson G, Bai AD, et al. Insulin pump use compared with intravenous insulin during labour and delivery: The INSPIRED observational cohort study. Diabet Med 2016;33:
- Fresa R, Visalli N, Di Blasi V, et al. Experiences of continuous subcutaneous insulin infusion in pregnant women with type 1 diabetes during delivery from four Italian centers: A retrospective observational study. Diabetes Technol Ther 2013;15:328–34.
- Cordua S, Secher AL, Ringholm L, et al. Real-time continuous glucose monitoring during labour and delivery in women with Type 1 diabetes—observations from a randomized controlled trial. Diabet Med 2013;30:1374–81.
- Ringholm L, Mathiesen ER, Kelstrup L, et al. Managing type 1 diabetes mellitus in pregnancy–from planning to breastfeeding. Nat Rev Endocrinol 2012;8:659–67.
- Roeder HA, Moore TR, Ramos GA. Changes in postpartum insulin requirements for patients with well-controlled type 1 diabetes. Am J Perinatol 2016;33:683–7.
- Achong N, Duncan EL, McIntyre HD, et al. Peripartum management of glycemia in women with type 1 diabetes. Diabetes Care 2014;37:364–71.
- Persaud RR, Azad MB, Chari RS, et al. Perinatal antibiotic exposure of neonates in Canada and associated risk factors: A population-based study. J Matern Fetal Neonatal Med 2015;28:1190–5.
- Davies HA, Clark JD, Dalton KJ, et al. Insulin requirements of diabetic women who breast feed. BMJ 1989;298:1357–8.
- Riviello C, Mello G, Jovanovic LG. Breastfeeding and the basal insulin requirement in type 1 diabetic women. Endocr Pract 2009;15:187–93.
- Stage E, Nørgård H, Damm P, et al. Long-term breast-feeding in women with type 1 diabetes. Diabetes Care 2006;29:771–4.
- Huang T, Brown FM, Curran A, et al. Association of pre-pregnancy BMI and postpartum weight retention with postpartum HbA1c among women with type 1 diabetes. Diabet Med 2015;32:181–8.
- Cyganek K, Hebda-Szydlo A, Skupien J, et al. Postpregnancy glycemic control and weight changes in type 1 diabetic women. Diabetes Care 2013;36:1083–7.
- Finkelstein SA, Keely E, Feig DS, et al. Breastfeeding in women with diabetes: Lower rates despite greater rewards. A population-based study. Diabet Med 2013;30:1094–101.
- Alvarez-Marfany M, Roman SH, Drexler AJ, et al. Long-term prospective study of postpartum thyroid dysfunction in women with insulin dependent diabetes mellitus. J Clin Endocrinol Metab 1994;79:10–16.
- Umpierrez GE, Latif KA, Murphy MB, et al. Thyroid dysfunction in patients with type 1 diabetes: A longitudinal study. Diabetes Care 2003;26:1181–5.
- Soltani H, Arden M. Factors associated with breastfeeding up to 6 months postpartum in mothers with diabetes. J Obstet Gynecol Neonatal Nurs 2009;38:586–94.
- Simmons D, Conroy C, Thompson CF. In-hospital breast feeding rates among women with gestational diabetes and pregestational Type 2 diabetes in South Auckland. Diabet Med 2005;22:177–81.
- Oza-Frank R, Chertok I, Bartley A. Differences in breast-feeding initiation and continuation by maternal diabetes status. Public Health Nutr 2015;18:727–35.
- Neubauer SH, Ferris AM, Chase CG, et al. Delayed lactogenesis in women with insulin-dependent diabetes mellitus. Am J Clin Nutr 1993;58:54–60.
- Ferris AM, Dalidowitz CK, Ingardia CM, et al. Lactation outcome in insulindependent diabetic women. J Am Diet Assoc 1988;88:317–22.
- Hummel S, Winkler C, Schoen S, et al. Breastfeeding habits in families with Type 1 diabetes. Diabet Med 2007;24:671–6.
- Hernandez TL, Anderson MA, Chartier-Logan C, et al. Strategies in the nutritional management of gestational diabetes. Clin Obstet Gynecol 2013;56:803–15.
- Riddle SW, Nommsen-Rivers LA. A case control study of diabetes during pregnancy and low milk supply. Breastfeed Med 2016;11:80–5.
- Schoen S, Sichert-HellertW, Hummel S, et al. Breastfeeding duration in families with type 1 diabetes compared to non-affected families: Results from BABYDIAB and DONALD studies in Germany. Breastfeed Med 2008;3:171–5.
- Sorkio S, Cuthbertson D, Barlund S, et al. Breastfeeding patterns of mothers with type 1 diabetes: Results from an infant feeding trial. Diabetes Metab Res Rev 2010;26:206–11.
- Benz J. Antidiabetic agents and lactation. J Hum Lact 1992;8:27–8.
- Ostrom KM, Ferris AM. Prolactin concentrations in serum and milk of mothers with and without insulin-dependent diabetes mellitus. Am J Clin Nutr 1993;58:49–53.
- Bromiker R, Rachamim A, Hammerman C, et al. Immature sucking patterns in infants of mothers with diabetes. J Pediatr 2006;149:640–3.
- Gardiner SJ, Kirkpatrick CM, Begg EJ, et al. Transfer of metformin into human milk. Clin Pharmacol Ther 2003;73:71–7.
- Briggs GG, Ambrose PJ, Nageotte MP, et al. Excretion of metformin into breast milk and the effect on nursing infants. Obstet Gynecol 2005;105:1437–41.
- Hale TW, Kristensen JH, Hackett LP, et al. Transfer of metformin into human milk. Diabetologia 2002;45:1509–14.
- Glueck CJ, Salehi M, Sieve L, et al. Growth, motor, and social development in breast- and formula-fed infants of metformin-treated women with polycystic ovary syndrome. J Pediatr 2006;148:628–32.
- Feig DS, Briggs GG, Kraemer JM, et al. Transfer of glyburide and glipizide into breast milk. Diabetes Care 2005;28:1851–5.
- Kulski JK, Hartmann PE. Milk insulin, GH and TSH: Relationship to changes in milk lactose, glucose and protein during lactogenesis in women. Endocrinol Exp 1983;17:317–26.
- Koldovský O. Hormones in milk. Vitam Horm 1995;50:77–149.
- Whitmore TJ, Trengove NJ, Graham DF, et al. Analysis of insulin in human breast milk in mothers with type 1 and type 2 diabetes mellitus. Int J Endocrinol 2012;2012:296368.
- Shehadeh N, Gelertner L, Blazer S, et al. Importance of insulin content in infant diet: Suggestion for a new infant formula. Acta Paediatr 2001;90:93–5.
- Shehadeh N, Shamir R, Berant M, et al. Insulin in human milk and the prevention of type 1 diabetes. Pediatr Diabetes 2001;2:175–7.
- Tiittanen M, Paronen J, Savilahti E, et al. Dietary insulin as an immunogen and tolerogen. Pediatr Allergy Immunol 2006;17:538–43.
- Cordero L, Ramesh S, Hillier K, et al. Early feeding and neonatal hypoglycemia in infants of diabeticmothers. Sage Open Med 2013;1:2050312113516613.
- Al Mamun A, O’Callaghan MJ, Williams GM, et al. Breastfeeding is protective to diabetes risk in young adults: A longitudinal study. Acta Diabetol 2015;52:837–44.
- Knip M, Akerblom HK, Becker D, et al. Hydrolyzed infant formula and early beta-cell autoimmunity: A randomized clinical trial. JAMA 2014;311:2279–87.
- Mayer-Davis EJ, Rifas-Shiman SL, Zhou L, et al. Breast-feeding and risk for childhood obesity: Does maternal diabetes or obesity status matter? Diabetes Care 2006;29:2231–7.
- Yan J, Liu L, Zhu Y, et al. The association between breastfeeding and childhood obesity: A meta-analysis. BMC Public Health 2014;14:1267.
- WHO Technical Staff. Exclusive breastfeeding to reduce the risk of childhood overweight and obesity. Biological, behavioural and contextual rationale. Geneva: World Health Organization, 2014. http://www.who.int/elena/bbc/breastfeeding_childhood_obesity/en/. Accessed January 2017.
- Health Canada, Canadian Paediatric Society, Dietitians of Canada & Breastfeeding Committee for Canada. Nutrition for healthly term infants: recommendations from six to 24 months. Ottawa, ON: Health Canada, 2014. https://www.canada.ca/en/health-canada/services/food-nutrition/healthy-eating/infant-feeding/nutrition-healthy-term-infants-recommendations-birth-sixmonths/6-24-months.html. Accessed January 2017.
- Critch JN, Canadian Paediatric Society Nutrition Gastroenterology Committee. Nutrition for healthy term infants, birth to six months: An overview. Paediatr Child Health 2013;18:206–7.
- Black A, Francoeur D, Rowe T, et al. SOGC clinical practice guidelines: Canadian contraception consensus. J Obstet Gynaecol Can 2004;26:219–96.
- Update to CDC’s U.S. medical eligibility criteria for contraceptive use, 2010: revised recommendations for the use of contraceptivemethods during the postpartum period. Atlanta: Centers for Disease Control and Prevention, 2011. http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6026a3.htm?s_cid=mm6026a3_w. Accessed January 2017.
- Guariguata L, Linnenkamp U, Beagley J, et al. Global estimates of the prevalence of hyperglycaemia in pregnancy. Diabetes Res Clin Pract 2014;103:176–85.
- Lowe WL Jr, Scholtens DM, Sandler V, et al. Genetics of gestational diabetes mellitus and maternal metabolism. Curr Diab Rep 2016;16:15.
- Stuebe AM, Wise A, Nguyen T, et al. Maternal genotype and gestational diabetes. Am J Perinatol 2014;31:69–76.
- Stanley K, Fraser R, Bruce C. Physiological changes in insulin resistance in human pregnancy: Longitudinal study with the hyperinsulinaemic euglycaemic clamp technique. Br J Obstet Gynaecol 1998;105:756–9.
- Madhuvrata P, Govinden G, Bustani R, et al. Prevention of gestational diabetes in pregnant women with risk factors for gestational diabetes: A systematic review and meta-analysis of randomised trials. Obstet Med 2015;8:68–85.
- Rogozinska E, Chamillard M, Hitman GA, et al. Nutritional manipulation for the primary prevention of gestational diabetes mellitus: A meta-analysis of randomised studies. PLoS ONE 2015;10:e0115526.
- Song C, Li J, Leng J, et al. Lifestyle intervention can reduce the risk of gestational diabetes: A meta-analysis of randomized controlled trials. Obes Rev 2016;17:960–9.
- Bao W, Bowers K, Tobias DK, et al. Prepregnancy dietary protein intake, major dietary protein sources, and the risk of gestational diabetes mellitus: A prospective cohort study. Diabetes Care 2013;36:2001–8.
- Bain E, Crane M, Tieu J, et al. Diet and exercise interventions for preventing gestational diabetes mellitus. Cochrane Database Syst Rev 2015;(4):CD010443.
- Russo LM, Nobles C, Ertel KA, et al. Physical activity interventions in pregnancy and risk of gestational diabetes mellitus: A systematic review and metaanalysis. Obstet Gynecol 2015;125:576–82.
- Chiswick C, Reynolds RM, Denison F, et al. Effect of metformin on maternal and fetal outcomes in obese pregnant women (EMPOWaR): A randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol 2015;3:778–86.
- Zhuo Z, Wang A, Yu H. Effect of metformin intervention during pregnancy on the gestational diabetes mellitus in women with polycystic ovary syndrome: A systematic review and meta-analysis. J Diabetes Res 2014;2014:381231.
- Harvey NC, Holroyd C, Ntani G, et al. Vitamin D supplementation in pregnancy: A systematic review. Health Technol Assess 2014;18:1–190.
- Lu M, Xu Y, Lv L, et al. Association between vitamin D status and the risk of gestational diabetes mellitus: A meta-analysis. Arch Gynecol Obstet 2016;293:959–66.
- Pérez-López FR, Pasupuleti V, Mezones-Holguin E, et al. Effect of vitamin D supplementation during pregnancy on maternal and neonatal outcomes: A systematic review and meta-analysis of randomized controlled trials. Fertil Steril 2015;103:1278–88, e4.
- Barrett HL, Callaway LK, Nitert MD. Probiotics: A potential role in the prevention of gestational diabetes? Acta Diabetol 2012;49:S1–13.
- Luoto R, Laitinen K, Nermes M, et al. Impact of maternal probiotic-supplemented dietary counselling on pregnancy outcome and prenatal and postnatal growth: A double-blind, placebo-controlled study. Br J Nutr 2010;103:1792–9.
- Lindsay KL, Brennan L, Kennelly MA, et al. Impact of probiotics in women with gestational diabetes mellitus on metabolic health: A randomized controlled trial. Am J Obstet Gynecol 2015;212:496, e1-11.
- Santamaria A, Di Benedetto A, Petrella E, et al. Myo-inositol may prevent gestational diabetes onset in overweight women: A randomized, controlled trial. J Matern Fetal Neonatal Med 2016;29:3234–7.
- D’Anna R, Di Benedetto A, Scilipoti A, et al. Myo-inositol supplementation for prevention of gestational diabetes in obese pregnant women: A randomized controlled trial. Obstet Gynecol 2015;126:310–15.
- Brunner S, Stecher L, Ziebarth S, et al. Excessive gestational weight gain prior to glucose screening and the risk of gestational diabetes: A meta-analysis. Diabetologia 2015;58:2229–37.
- McBain RD, Dekker GA, Clifton VL, et al. Impact of inter-pregnancy BMI change on perinatal outcomes: A retrospective cohort study. Eur J Obstet Gynecol Reprod Biol 2016;205:98–104.
- Galazis N, Docheva N, Simillis C, et al. Maternal and neonatal outcomes in women undergoing bariatric surgery: A systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol 2014;181:45–53.
- American College of Obstetricians and Gynecologists. ACOG practice bulletin no. 105: Bariatric surgery and pregnancy. Obstet Gynecol 2009;113:1405–13.
- Mahawar KK, Graham Y, Small PK. Optimum time for pregnancy after bariatric surgery. Surg Obes Relat Dis 2016;12:1126–8.
- Smith J, Cianflone K, Biron S, et al. Effects of maternal surgical weight loss in mothers on intergenerational transmission of obesity. J Clin Endocrinol Metab 2009;94:4275–83.
- Riskin-Mashiah S, Younes G, Damti A, et al. First-trimester fasting hyperglycemia and adverse pregnancy outcomes. Diabetes Care 2009;32:1639–43.
- Zhu W, Yang H,Wei Y, et al. Evaluation of the value of fasting plasma glucose in the first prenatal visit to diagnose gestational diabetes mellitus in China. Diabetes Care 2013;36:586–90.
- Rowan JA, Budden A, Ivanova V, et al. Women with an HbA1c of 41–49 mmol/mol (5.9–6.6%): A higher risk subgroup that may benefit from early pregnancy intervention. Diabet Med 2016;33:25–31.
- Osmundson SS, Zhao BS, Kunz L, et al. First trimester hemoglobin A1c prediction of gestational diabetes. Am J Perinatol 2016;33:977–82.
- Mañé L, Flores-Le Roux JA, Benaiges D, et al. Role of first trimester HbA1c as a predictor of adverse obstetric outcomes in a multi-ethnic cohort. J Clin Endocrinol Metab 2017;102:390–7.
- Granada C, Forbes J, Sangi-Haghpeykar H, et al. Can overt diabetes mellitus be predicted by an early A1C value in gestational diabetics? J Reprod Med 2014;59:343–7.
- Alunni ML, Roeder HA, Moore TR, et al. First trimester gestational diabetes screening—change in incidence and pharmacotherapy need. Diabetes Res Clin Pract 2015;109:135–40.
- Sweeting AN, Ross GP, Hyett J, et al. Gestational diabetes mellitus in early pregnancy: Evidence for poor pregnancy outcomes despite treatment. Diabetes Care 2016;39:75–81.
- Wong T, Ross GP, Jalaludin BB, et al. The clinical significance of overt diabetes in pregnancy. Diabet Med 2013;30:468–74.
- Crowther CA, Hiller JE, Moss JR, et al. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med 2005;352:2477–86.
- Hartling L, Dryden DM, Guthrie A, et al. Benefits and harms of treating gestational diabetes mellitus: A systematic review and meta-analysis for the U.S. Preventive Services Task Force and the National Institutes of Health Office of Medical Applications of Research. Ann Intern Med 2013;159:123–9.
- Horvath K, Koch K, Jeitler K, et al. Effects of treatment in women with gestational diabetes mellitus: Systematic review and meta-analysis. BMJ 2010;340:c1395.
- Falavigna M, Schmidt MI, Trujillo J, et al. Effectiveness of gestational diabetes treatment: A systematic review with quality of evidence assessment. Diabetes Res Clin Pract 2012;98:396–405.
- Canadian Diabetes Association Clinical Practice Guidelines Expert Committee. Canadian Diabetes Association 2013 clinical practice guidelines for the prevention and management of diabetes in Canada. Can J Diabetes 2013;37:S1–212.
- Agarwal MM, Dhatt GS, Punnose J, et al. Gestational diabetes in a high-risk population: Using the fasting plasma glucose to simplify the diagnostic algorithm. Eur J Obstet Gynecol Reprod Biol 2005;120:39–44.
- Agarwal MM, Dhatt GS. Fasting plasma glucose as a screening test for gestational diabetes mellitus. Arch Gynecol Obstet 2007;275:81–7.
- Agarwal MM, Dhatt GS, Othman Y, et al. Gestational diabetes: Fasting capillary glucose as a screening test in a multi-ethnic, high-risk population. Diabet Med 2009;26:760–5.
- AltoWA. No need for glycosuria/proteinuria screen in pregnant women. J Fam Pract 2005;54:978–83.
- Fadl H, Ostlund I, Nilsson K, et al. Fasting capillary glucose as a screening test for gestational diabetes mellitus. BJOG 2006;113:1067–71.
- Sacks DA, Chen W, Wolde-Tsadik G, et al. Fasting plasma glucose test at the first prenatal visit as a screen for gestational diabetes. Obstet Gynecol 2003;101:1197–203.
- Soumya S, Rohilla M, Chopra S, et al. HbA1c: A useful screening test for gestational diabetes mellitus. Diabetes Technol Ther 2015;17:899–904.
- Sermer M, Naylor CD, Gare DJ, et al. Impact of increasing carbohydrate intolerance on maternal-fetal outcomes in 3637 women without gestational diabetes. The Toronto Tri-Hospital Gestational Diabetes Project. Am J Obstet Gynecol 1995;173:146–56.
- Donovan LE, Savu A, Edwards AL, et al. Prevalence and timing of screening and diagnostic testing for gestational diabetes mellitus: A population-based study in Alberta, Canada. Diabetes Care 2016;39:55–60.
- Temming LA, Tuuli MG, Stout MJ, et al. Diagnostic ability of elevated 1-h glucose challenge test. J Perinatol 2016;36:342–6.
- Cheng YW, Esakoff TF, Block-Kurbisch I, et al. Screening or diagnostic: Markedly elevated glucose loading test and perinatal outcomes. J Matern Fetal Neonatal Med 2006;19:729–34.
- Hillier TA, Ogasawara KK, Pedula KL, et al. Markedly different rates of incident insulin treatment based on universal gestational diabetes mellitus screening in a diverse HMO population. Am J Obstet Gynecol 2013;209:440, e1-9.
- HAPO Study Cooperative Research Group, Metzger BE, Lowe LP, et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 2008;358:1991–2002.
- Sacks DA, Greenspoon JS, Abu-Fadil S, et al. Toward universal criteria for gestational diabetes: The 75-gram glucose tolerance test in pregnancy. Am J Obstet Gynecol 1995;172:607–14.
- VanDorsten JP, Dodson WC, Espeland MA, et al. NIH consensus development conference: Diagnosing gestational diabetes mellitus. NIH Consens State Sci Statements 2013;29:1–31.
- American Diabetes Assosication. 2. Classification and diagnosis of diabetes. Diabetes Care 2016;39:S13–22.
- Committee on Practice Bulletins–Obstetrics. Practice Bulletin No. 137: Gestational diabetes mellitus. Obstet Gynecol 2013;122:406–16.
- NICE. Diabetes in pregnancy: management from preconception to the postnatal period. London, UK: National Institute for Health and Care Excellence (NICE), 2015. https://www.nice.org.uk/guidance/ng3. Accessed January 2017.
- Hod M, Kapur A, Sacks DA, et al. The International Federation of Gynecology and Obstetrics (FIGO) Initiative on gestational diabetes mellitus: A pragmatic guide for diagnosis, management, and care. Int J Gynaecol Obstet 2015;131(Suppl. 3):S173–211.
- Nankervis W, McIntyre HD, Moses R, et al. ADIPS consensus guidelines for the testing and diagnosis of hyperglycaemia in pregnancy in Australia and New Zealand. Royal College of Pathologists of Australasia: Australasian Diabetes in Pregnancy Society, 2014. http://adips.org/downloads/2014ADIPSGDMGuidelines V18.11.2014_000.pdf. Accessed January 2017.
- Sacks DA, Black MH, Li X, et al. Adverse pregnancy outcomes using the International Association of the Diabetes and Pregnancy Study Groups criteria: Glycemic thresholds and associated risks. Obstet Gynecol 2015;126:67–73.
- Wu ET, Nien FJ, Kuo CH, et al. Diagnosis of more gestational diabetes lead to better pregnancy outcomes: Comparing the International Association of the Diabetes and Pregnancy study group criteria, and the Carpenter and Coustan criteria. J Diabetes Investig 2016;7:121–6.
- Kong JM, Lim K, Thompson DM. Evaluation of the International Association of the Diabetes In Pregnancy Study Group newcriteria: Gestational diabetes project. Can J Diabetes 2015;39:128–32.
- Bodmer-Roy S, Morin L, Cousineau J, et al. Pregnancy outcomes in women with and without gestational diabetes mellitus according to the International Association of the Diabetes and Pregnancy Study Groups criteria. Obstet Gynecol 2012;120:746–52.
- Duran A, Saenz S, Torrejon MJ, et al. Introduction of IADPSG criteria for the screening and diagnosis of gestational diabetes mellitus results in improved pregnancy outcomes at a lower cost in a large cohort of pregnant women: The St. Carlos Gestational Diabetes Study. Diabetes Care 2014;37:2442–50.
- Feldman RK, Tieu RS, Yasumura L. Gestational diabetes screening: The International Association of the Diabetes and Pregnancy study groups compared with Carpenter-Coustan screening. Obstet Gynecol 2016;127:10–17.
- Ogunleye OK, Davidson KD, Gregg AR, et al. Perinatal outcomes after adopting 1- versus 2-step approach to diagnosing gestational diabetes. J Matern Fetal Neonatal Med 2016;30:186–90.
- Coop C, Edlin R, Brown J, et al. Cost-effectiveness of the New Zealand diabetes in pregnancy guideline screening recommendations. BMJ Open 2015;5:e006996.
- Mission JF, Ohno MS, Cheng YW, et al. Gestational diabetes screening with the new IADPSG guidelines: A cost-effectiveness analysis. Am J Obstet Gynecol 2012;207:326, e1-9.
- Werner EF, Pettker CM, Zuckerwise L, et al. Screening for gestational diabetes mellitus: Are the criteria proposed by the international association of the Diabetes and Pregnancy Study groups cost-effective? Diabetes Care 2012;35:529–35.
- Kgosidialwa O, Egan AM, Carmody L, et al. Treatment with diet and exercise for women with gestational diabetes mellitus diagnosed using IADPSG criteria. J Clin Endocrinol Metab 2015;100:4629–36.
- Casey BM, Mele L, Landon MB, et al. Does maternal body mass index influence treatment effect in women with mild gestational diabetes? Am J Perinatol 2015;32:93–100.
- Farrar D, Duley L, Medley N, et al. Different strategies for diagnosing gestational diabetes to improve maternal and infant health. Cochrane Database Syst Rev 2015;(1):CD007122.
- Brahm AJ, Wang G, Wang J, et al. Genetic confirmation rate in clinically suspected maturity-onset diabetes of the young. Can J Diabetes 2016;40:555–60.
- Lachance CH, Baillargeon M. Should the clinical criteria for suspecting glucokinase mutation-related hyperglycemia (MODY-2) be revisited during pregnancy?. Can J Diabetes 2017 (in press).
- Wong T, Barnes RA, Ross GP, et al. Are the Institute of Medicine weight gain targets applicable in women with gestational diabetes mellitus? Diabetologia 2016;60:416–23.
- Harper LM, Tita A, Biggio JR. The institute of medicine guidelines for gestational weight gain after a diagnosis of gestational diabetes and pregnancy outcomes. Am J Perinatol 2015;32:239–46.
- Barquiel B, Herranz L, Hillman N, et al. HbA1c and gestational weight gain are factors that influence neonatal outcome in mothers with gestational diabetes. J Womens Health (Larchmt) 2016;25:579–85.
- Kim SY, Sharma AJ, Sappenfield W, et al. Association of maternal body mass index, excessive weight gain, and gestational diabetes mellitus with large-forgestational-age births. Obstet Gynecol 2014;123:737–44.
- American College of Obstetricians and Gynecologists. ACOG Committee opinion no. 548:Weight gain during pregnancy. Obstet Gynecol 2013;121:210–12.
- Catalano PM, Mele L, Landon MB, et al. Inadequate weight gain in overweight and obese pregnant women: What is the effect on fetal growth? Am J Obstet Gynecol 2014;211:137, e1-7.
- Katon J, Reiber G, Williams MA, et al. Weight loss after diagnosis with gestational diabetes and birth weight among overweight and obese women. Matern Child Health J 2013;17:374–83.
- Muktabhant B, Lawrie TA, Lumbiganon P, et al. Diet or exercise, or both, for preventing excessive weight gain in pregnancy. Cochrane Database Syst Rev 2015;(6):CD007145.
- Canadian Diabetes Association, Dietitians of Canada, Diabète Québec, et al. Recommendations for nutrition best practice in the management of gestational diabetes mellitus. Executive summary (1). Can J Diet Pract Res 2006;67:206–8.
- Fagen C, King JD, Erick M. Nutrition management in women with gestational diabetes mellitus: A review by ADA’s Diabetes Care and Education Dietetic Practice Group. J Am Diet Assoc 1995;95:460–7.
- Morisset AS, Côté JA, Michaud A, et al. Dietary intakes in the nutritional management of gestational diabetes mellitus. Can J Diet Pract Res 2014;75:64–71.
- Franz MJ, Bantle JP, Beebe CA, et al. Evidence-based nutrition principles and recommendations for the treatment and prevention of diabetes and related complications. Diabetes Care 2002;25:148–98.
- Jovanovic L. Medical nutritional therapy in pregnant women with pregestational diabetes mellitus. J Matern Fetal Med 2000;9:21–8.
- Dornhorst A, Frost G. The principles of dietary management of gestational diabetes: Reflection on current evidence. J Hum Nutr Diet 2002;15:145–56. quiz 57-9.
- Metzger BE, Buchanan TA, Coustan DR, et al. Summary and recommendations of the Fifth International Workshop-Conference on Gestational Diabetes Mellitus. Diabetes Care 2007;30(Suppl. 2):S251–60.
- Moreno-Castilla C, Mauricio D, HernandezM. Role of medical nutrition therapy in the management of gestational diabetes mellitus. Curr Diab Rep 2016;16:22.
- Jovanovic L. Achieving euglycaemia in women with gestational diabetes mellitus: Current options for screening, diagnosis and treatment. Drugs 2004;64:1401–17.
- Moses RG, Luebcke M, DavisWS, et al. Effect of a low-glycemic-index diet during pregnancy on obstetric outcomes. Am J Clin Nutr 2006;84:807–12.
- Han S, Crowther CA, Middleton P, et al. Different types of dietary advice for women with gestational diabetes mellitus. Cochrane Database Syst Rev 2013;(3):CD009275.
- Viana LV, Gross JL, Azevedo MJ. Dietary intervention in patients with gestational diabetes mellitus: A systematic review and meta-analysis of randomized clinical trials on maternal and newborn outcomes. Diabetes Care 2014;37:3345–55.
- Wei J, Heng W, Gao J. Effects of low glycemic index diets on gestational diabetes mellitus: A meta-analysis of randomized controlled clinical trials. Medicine (Baltimore) 2016;95:e3792.
- Lassi ZS, Bhutta ZA. Risk factors and interventions related to maternal and pre-pregnancy obesity, pre-diabetes and diabetes for maternal, fetal and neonatal outcomes: A systematic review. Expert Rev Obstet Gynecol 2013;8:639–60.
- Grant SM,Wolever TM, O’Connor DL, et al. Effect of a low glycaemic index diet on blood glucose in women with gestational hyperglycaemia. Diabetes Res Clin Pract 2011;91:15–22.
- Hu ZG, Tan RS, Jin D, et al. A low glycemic index staple diet reduces postprandial glucose values in Asian women with gestational diabetes mellitus. J Investig Med 2014;62:975–9.
- Louie JC, Brand-Miller JC, Moses RG. Carbohydrates, glycemic index, and pregnancy outcomes in gestational diabetes. Curr Diab Rep 2013;13:6–11.
- Institute of Medicine. Dietary reference intakes: energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids (macronutrients).Washington, DC: Institute of Medicine of the National Academics, 2005 http://www.nap.edu/read/10490/chapter/1. Accessed January 2017.
- Ruchat SM, Mottola MF. The important role of physical activity in the prevention and management of gestational diabetes mellitus. Diabetes Metab Res Rev 2013;29:334–46.
- Anjana RM, Sudha V, Lakshmipriya N, et al. Physical activity patterns and gestational diabetes outcomes—The wings project. Diabetes Res Clin Pract 2016;116:253–62.
- Harmon KA, Gerard L, Jensen DR, et al. Continuous glucose profiles in obese and normal-weight pregnant women on a controlled diet: Metabolic determinants of fetal growth. Diabetes Care 2011;34:2198–204.
- Rowan JA, Gao W, Hague WM, et al. Glycemia and its relationship to outcomes in the metformin in gestational diabetes trial. Diabetes Care 2010;33:9–16.
- Prutsky GJ, Domecq JP,Wang Z, et al. Glucose targets in pregnant women with diabetes: A systematic review and meta-analysis. J Clin Endocrinol Metab 2013;98:4319–24.
- Buchanan TA, Kjos SL, Montoro MN, et al. Use of fetal ultrasound to select metabolic therapy for pregnancies complicated by mild gestational diabetes. Diabetes Care 1994;17:275–83.
- Schaefer-Graf UM, Kjos SL, Fauzan OH, et al. A randomized trial evaluating a predominantly fetal growth-based strategy to guide management of gestational diabetes in Caucasian women. Diabetes Care 2004;27:297–302.
- Bonomo M, Cetin I, Pisoni MP, et al. Flexible treatment of gestational diabetes modulated on ultrasound evaluation of intrauterine growth: A controlled randomized clinical trial. Diabetes Metab 2004;30:237–44.
- Kjos SL, Schaefer-Graf U, Sardesi S, et al. A randomized controlled trial using glycemic plus fetal ultrasound parameters versus glycemic parameters to determine insulin therapy in gestational diabetes with fasting hyperglycemia. Diabetes Care 2001;24:1904–10.
- Balsells M, Garcia-Patterson A, Gich I, et al. Ultrasound-guided compared to conventional treatment in gestational diabetes leads to improved birthweight but more insulin treatment: Systematic review and meta-analysis. Acta Obstet Gynecol Scand 2014;93:144–51.
- Langer O, Yogev Y, Most O, et al. Gestational diabetes: The consequences of not treating. Am J Obstet Gynecol 2005;192:989–97.
- Hawkins JS, Casey BM, Lo JY, et al.Weekly compared with daily blood glucose monitoring in women with diet-treated gestational diabetes. Obstet Gynecol 2009;113:1307–12.
- Kestila KK, Ekblad UU, Ronnemaa T. Continuous glucose monitoring versus selfmonitoring of blood glucose in the treatment of gestational diabetes mellitus. Diabetes Res Clin Pract 2007;77:174–9.
- Cypryk K, Pertyn´ ska-Marczewska M, Szymczak W, et al. Evaluation of metabolic control in women with gestational diabetes mellitus by the continuous glucose monitoring system: A pilot study. Endocr Pract 2006;12:245–50.
- McLachlan K, Jenkins A, O’Neal D. The role of continuous glucose monitoring in clinical decision-making in diabetes in pregnancy. Aust N Z J Obstet Gynaecol 2007;47:186–90.
- Yu F, Lv L, Liang Z, et al. Continuous glucose monitoring effects on maternal glycemic control and pregnancy outcomes in patients with gestational diabetes mellitus: A prospective cohort study. J Clin Endocrinol Metab 2014;99:4674–82.
- Wei Q, Sun Z, Yang Y, et al. Effect of a CGMS and SMBG on maternal and neonatal outcomes in gestational diabetes mellitus: A randomized controlled trial. Sci Rep 2016;6:19920.
- Ornoy A, Ratzon N, Greenbaum C, et al. Neurobehaviour of school age children born to diabetic mothers. Arch Dis Child Fetal Neonatal Ed 1998;79:F94–9.
- Carral F, Ayala Mdel C, Fernández JJ, et al. Web-based telemedicine system is useful for monitoring glucose control in pregnant women with diabetes. Diabetes Technol Ther 2015;17:349–54.
- Perez-Ferre N, Galindo M, Fernandez MD, et al. A Telemedicine system based on Internet and shortmessage service as a new approach in the follow-up of patients with gestational diabetes. Diabetes Res Clin Pract 2010;87:e15–17.
- Homko CJ, Santamore WP, Whiteman V, et al. Use of an internet-based telemedicine system to manage underserved women with gestational diabetes mellitus. Diabetes Technol Ther 2007;9:297–306.
- Kruger DF, White K, Galpern A, et al. Effect of modem transmission of blood glucose data on telephone consultation time, clinic work flow, and patient satisfaction for patients with gestational diabetes mellitus. J Am Acad Nurse Pract 2003;15:371–5.
- Homko CJ, Deeb LC, Rohrbacher K, et al. Impact of a telemedicine system with automated reminders on outcomes in women with gestational diabetes mellitus. Diabetes Technol Ther 2012;14:624–9.
- Perez-Ferre N, Galindo M, Fernandez MD, et al. The outcomes of gestational diabetes mellitus after a telecare approach are not inferior to traditional outpatient clinic visits. Int J Endocrinol 2010;2010:386941.
- Wojcicki JM, Ladyzynski P, Krzymien J, et al. What we can really expect from telemedicine in intensive diabetes treatment: Results from 3-year study on type 1 pregnant diabetic women. Diabetes Technol Ther 2001;3:581–9.
- Frost D, Beischer W. Telemedicine in the management of pregnancy in type 1 diabetic women. Diabetes Care 2000;23:863–4.
- Dalfra MG, Nicolucci A, Lapolla A. The effect of telemedicine on outcome and quality of life in pregnant women with diabetes. J Telemed Telecare 2009;15:238–42.
- Mastrogiannis DS, Igwe E, Homko CJ. The role of telemedicine in the management of the pregnancy complicated by diabetes. Curr Diab Rep 2013;13:1–5.
- Poorman E, Gazmararian J, Parker RM, et al. Use of text messaging for maternal and infant health: A systematic review of the literature. Matern Child Health J 2015;19:969–89.
- O’Brien OA, McCarthy M, Gibney ER, et al. Technology-supported dietary and lifestyle interventions in healthy pregnant women: A systematic review. Eur J Clin Nutr 2014;68:760–6.
- Hirst JE, Mackillop L, Loerup L, et al. Acceptability and user satisfaction of a smartphone-based, interactive blood glucose management system in women with gestational diabetes mellitus. J Diabetes Sci Technol 2015;9:111–15.
- Rasekaba TM, Furler J, Blackberry I, et al. Telemedicine interventions for gestational diabetes mellitus: A systematic review and meta-analysis. Diabetes Res Clin Pract 2015;110:1–9.
- Langer O. Management of gestational diabetes: Pharmacologic treatment options and glycemic control. Endocrinol Metab Clin North Am 2005;35:53–78.
- Hadden DR. When and how to start insulin treatment in gestational diabetes: A UK perspective. Diabet Med 2001;18:960–4.
- Mecacci F, Carignani L, Cioni R, et al. Maternal metabolic control and perinatal outcome in women with gestational diabetes treated with regular or lispro insulin: Comparison with non-diabetic pregnant women. Eur J Obstet Gynecol Reprod Biol 2003;111:19–24.
- Pettitt DJ, Ospina P, Kolaczynski JW, et al. Comparison of an insulin analog, insulin aspart, and regular human insulin with no insulin in gestational diabetes mellitus. Diabetes Care 2003;26:183–6.
- Pettitt DJ, Ospina P, Howard C, et al. Efficacy, safety and lack of immunogenicity of insulin aspart compared with regular human insulin for women with gestational diabetes mellitus. Diabet Med 2007;24:1129–35.
- Giri H, Chandel S, Dwarakanath LS, et al. Increased endothelial inflammation, sTie-2 and arginase activity in umbilical cords obtained from gestational diabetic mothers. PLoS ONE 2013;8:e84546.
- Zhao LP, Sheng XY, Zhou S, et al. Metformin versus insulin for gestational diabetes mellitus: A meta-analysis. Br J Clin Pharmacol 2015;80:1224–34.
- Jiang YF, Chen XY, Ding T, et al. Comparative efficacy and safety of OADs in management of GDM: Network meta-analysis of randomized controlled trials. J Clin Endocrinol Metab 2015;100:2071–80.
- Balsells M, Garcia-Patterson A, Sola I, et al. Glibenclamide, metformin, and insulin for the treatment of gestational diabetes: A systematic review and metaanalysis. BMJ 2015;350:h102.
- Poolsup N, Suksomboon N, Amin M. Efficacy and safety of oral antidiabetic drugs in comparison to insulin in treating gestational diabetes mellitus: A metaanalysis. PLoS ONE 2014;9:e109985.
- Kitwitee P, Limwattananon S, Limwattananon C, et al. Metformin for the treatment of gestational diabetes: An updated meta-analysis. Diabetes Res Clin Pract 2015;109:521–32.
- Feng Y, Yang H. Metformin—a potentially effective drug for gestational diabetes mellitus: A systematic review and meta-analysis. J Matern Fetal Neonatal Med 2016;9:1–8.
- Rowan JA, Hague WM, Gao W, et al. Metformin versus insulin for the treatment of gestational diabetes. N Engl J Med 2008;358:2003–15.
- Rowan JA, Rush EC, Obolonkin V, et al. Metformin in gestational diabetes: The offspring follow-up (MiG TOFU): Body composition at 2 years of age. Diabetes Care 2011;34:2279–84.
- Ijäs H, Vääräsmäki M, Saarela T, et al. A follow-up of a randomised study of metformin and insulin in gestational diabetes mellitus: Growth and development of the children at the age of 18 months. BJOG 2015;122:994–1000.
- Tertti K, Eskola E, Ronnemaa T, et al. Neurodevelopment of two-year-old children exposed to metformin and insulin in gestational diabetes mellitus. J Dev Behav Pediatr 2015;36:752–7.
- Wouldes TA, Battin M, Coat S, et al. Neurodevelopmental outcome at 2 years in offspring of women randomised to metformin or insulin treatment for gestational diabetes. Arch Dis Child Fetal Neonatal Ed 2016 (in press).
- Bertini AM, Silva JC, Taborda W, et al. Perinatal outcomes and the use of oral hypoglycemic agents. J Perinat Med 2005;33:519–23.
- Obenshain SS, Adam PA, King KC, et al. Human fetal insulin response to sustained maternal hyperglycemia. N Engl J Med 1970;283:566–70.
- Lucas A, Morley R, Cole TJ. Adverse neurodevelopmental outcome of moderate neonatal hypoglycaemia. BMJ 1988;297:1304–8.
- Koivisto M, Blanco-Sequeiros M, Krause U. Neonatal symptomatic and asymptomatic hypoglycaemia: A follow-up study of 151 children. Dev Med Child Neurol 1972;14:603–14.
- Stenninger E, Flink R, Eriksson B, et al. Long-term neurological dysfunction and neonatal hypoglycaemia after diabetic pregnancy. Arch Dis Child Fetal Neonatal Ed 1998;79:F174–9.
- Singhi S. Effect of maternal intrapartum glucose therapy on neonatal blood glucose levels and neurobehavioral status of hypoglycemic term newborn infants. J Perinat Med 1988;16:217–24.
- Kenepp NB, Kumar S, Shelley WC, et al. Fetal and neonatal hazards of maternal hydration with 5% dextrose before caesarean section. Lancet 1982;1:1150–2.
- Miodovnik M, Mimouni F, Tsang RC, et al. Management of the insulindependent diabetic during labor and delivery. Influences on neonatal outcome. Am J Perinatol 1987;4:106–14.
- Andersen O, Hertel J, Schmølker L, et al. Influence of the maternal plasma glucose concentration at delivery on the risk of hypoglycaemia in infants of insulindependent diabetic mothers. Acta Paediatr Scand 1985;74:268–73.
- Curet LB, Izquierdo LA, Gilson GJ, et al. Relative effects of antepartum and intrapartum maternal blood glucose levels on incidence of neonatal hypoglycemia. J Perinatol 1997;17:113–15.
- Lean ME, Pearson DW, Sutherland HW. Insulin management during labour and delivery in mothers with diabetes. Diabet Med 1990;7:162–4.
- Feldberg D, Dicker D, Samuel N, et al. Intrapartum management of insulindependent diabetes mellitus (IDDM) gestants. a comparative study of constant intravenous insulin infusion and continuous subcutaneous insulin infusion pump (CSIIP). Acta Obstet Gynecol Scand 1988;67:333–8.
- Stenninger E, Lindqvist A, Aman J, et al. Continuous Subcutaneous Glucose Monitoring System in diabetic mothers during labour and postnatal glucose adaptationof their infants. Diabet Med 2008;25:450–4.
- Balsells M, Corcoy R, Adelantado JM, et al. Gestational diabetes mellitus: Metabolic control during labour. Diabetes Nutr Metab 2000;13:257–62.
- Golde SH, Good-Anderson B, Montoro M, et al. Insulin requirements during labor: A reappraisal. Am J Obstet Gynecol 1982;144:556–9.
- Jovanovic L, Peterson CM. Insulin and glucose requirements during the first stage of labor in insulin-dependent diabetic women. Am J Med 1983;75:607–12.
- Carron Brown S, Kyne-Grzebalski D,Mwangi B, et al. Effect of management policy upon 120 Type 1 diabetic pregnancies: Policy decisions in practice. Diabet Med 1999;16:573–8.
- Barrett HL, Morris J, McElduff A. Watchful waiting: A management protocol for maternal glycaemia in the peripartum period. Aust N Z J Obstet Gynaecol 2009;49:162–7.
- Rosenberg VA, Eglinton GS, Rauch ER, et al. Intrapartum maternal glycemic control in women with insulin requiring diabetes: A randomized clinical trial of rotating fluids versus insulin drip. Am J Obstet Gynecol 2006;195:1095–9.
- Schaefer-Graf UM, Hartmann R, Pawliczak J, et al. Association of breastfeeding and early childhood overweight in children from mothers with gestational diabetes mellitus. Diabetes Care 2006;29:1105–7.
- Stuebe AM, Rich-Edwards JW, WillettWC, et al. Duration of lactation and incidence of type 2 diabetes. JAMA 2005;294:2601–10.
- Gunderson EP, Jacobs DR Jr, Chiang V, et al. Duration of lactation and incidence of the metabolic syndrome in women of reproductive age according to gestational diabetes mellitus status: A 20-Year prospective study in CARDIA (Coronary Artery Risk Development in Young Adults). Diabetes 2010;59:495–504.
- Liu B, Jorm L, Banks E. Parity, breastfeeding, and the subsequent risk of maternal type 2 diabetes. Diabetes Care 2010;33:1239–41.
- Feig DS, Lipscombe LL, Tomlinson G, et al. Breastfeeding predicts the risk of childhood obesity in a multi-ethnic cohort of women with diabetes. J Matern Fetal Neonatal Med 2011;24:511–15.
- Gunderson EP, Hurston SR, Ning X, et al. Lactation and progression to type 2 diabetes mellitus after gestational diabetes mellitus: A prospective cohort study. Ann Intern Med 2015;163:889–98.
- Aune D, Norat T, Romundstad P, et al. Breastfeeding and the maternal risk of type 2 diabetes: A systematic review and dose-responsemeta-analysis of cohort studies. Nutr Metab Cardiovasc Dis 2014;24:107–15.
- Chowdhury R, Sinha B, Sankar MJ, et al. Breastfeeding and maternal health outcomes: A systematic review and meta-analysis. Acta Paediatr 2015;104:96–113.
- Much D, Beyerlein A, Roßbauer M, et al. Beneficial effects of breastfeeding in women with gestational diabetes mellitus. Mol Metab 2014;3:284–92.
- Morrison MK, Collins CE, Lowe JM, et al. Factors associated with early cessation of breastfeeding inwomen with gestational diabetes mellitus.Women Birth 2015;28:143–7.
- Catalano PM, Drago NM, Amini SB. Longitudinal changes in pancreatic betacell function and metabolic clearance rate of insulin in pregnant women with normal and abnormal glucose tolerance. Diabetes Care 1998;21:403–8.
- Ergin T, Lembet A, Duran H, et al. Does insulin secretion in patients with one abnormal glucose tolerance test value mimic gestational diabetes mellitus? Am J Obstet Gynecol 2002;186:204–9.
- Kjos SL, Peters RK, Xiang A, et al. Predicting future diabetes in Latino women with gestational diabetes. Utility of early postpartum glucose tolerance testing. Diabetes 1995;44:586–91.
- Pallardo F, Herranz L, Garcia-Ingelmo T, et al. Early postpartum metabolic assessment in women with prior gestational diabetes. Diabetes Care 1999;22:1053–8.
- O’Sullivan JB. Diabetes mellitus after GDM. Diabetes 1991;40:131–5.
- Kim C, Newton KM, Knopp RH. Gestational diabetes and the incidence of type 2 diabetes: A systematic review. Diabetes Care 2002;25:1862–8.
- Kaufmann RC, Schleyhahn FT, Huffman DG, et al. Gestational diabetes diagnostic criteria: Long-term maternal follow-up. Am J Obstet Gynecol 1995;172:621–5.
- Schaefer-Graf UM, Buchanan TA, Xiang AH, et al. Clinical predictors for a high risk for the development of diabetes mellitus in the early puerperium in women with recent gestational diabetes mellitus. Am J Obstet Gynecol 2002;186:751–6.
- Rayanagoudar G, Hashi AA, Zamora J, et al. Quantification of the type 2 diabetes risk in women with gestational diabetes: A systematic review and metaanalysis of 95,750 women. Diabetologia 2016;59:1403–11.
- Ekelund M, Shaat N, Almgren P, et al. Prediction of postpartum diabetes in women with gestational diabetes mellitus. Diabetologia 2010;53:452–7.
- Cheung NW, Helmink D. Gestational diabetes: The significance of persistent fasting hyperglycemia for the subsequent development of diabetes mellitus. J Diabetes Complications 2006;20:21–5.
- Oldfield MD, Donley P, Walwyn L, et al. Long term prognosis of women with gestational diabetes in a multiethnic population. Postgrad Med J 2007;83:426–30.
- Retnakaran R, Shah BR. Abnormal screening glucose challenge test in pregnancy and future risk of diabetes in young women. Diabet Med 2009;26:474–7.
- Feig DS, Shah BR, Lipscombe LL, et al. Preeclampsia as a risk factor for diabetes: A population-based cohort study. PLoS Med 2013;10:e1001425.
- Järvelä IY, Juutinen J, Koskela P, et al. Gestational diabetes identifies women at risk for permanent type 1 and type 2 diabetes in fertile age: Predictive role of autoantibodies. Diabetes Care 2006;29:607–12.
- Dozio N, Beretta A, Belloni C, et al. Low prevalence of islet autoantibodies in patients with gestational diabetes mellitus. Diabetes Care 1997;20:81–3.
- Löbner K, Knopff A, Baumgarten A, et al. Predictors of postpartum diabetes in women with gestational diabetes mellitus. Diabetes 2006;55:792–7.
- Smirnakis KV, Chasan-Taber L, Wolf M, et al. Postpartum diabetes screening inwomenwith a history of gestational diabetes. Obstet Gynecol 2005;106:1297– 303.
- Clark HD, van Walraven C, Code C, et al. Did publication of a clinical practice guideline recommendation to screen for type 2 diabetes in women with gestational diabetes change practice? Diabetes Care 2003;26:265–8.
- Morrison MK, Lowe JM, Collins CE. Perceived risk of Type 2 diabetes in Australian women with a recent history of gestational diabetes mellitus. Diabet Med 2010;27:882–6.
- Kerimoglu OS, Yalvac S, Karcaaltincaba D, et al. Early post-partum diabetes mellitus screening rates in patients with history of gestational diabetes. Arch Gynecol Obstet 2010;282:613–16.
- Kim C, Tabaei BP, Burke R, et al. Missed opportunities for type 2 diabetes mellitus screening among women with a history of gestational diabetes mellitus. Am J Public Health 2006;96:1643–8.
- Russell MA, Phipps MG, Olson CL, et al. Rates of postpartum glucose testing after gestational diabetes mellitus. Obstet Gynecol 2006;108:1456–62.
- McGovern A, Butler L, Jones S, et al. Diabetes screening after gestational diabetes in England: A quantitative retrospective cohort study. Br J Gen Pract 2014;64:e17–23.
- Middleton P, Crowther CA. Reminder systems for women with previous gestational diabetes mellitus to increase uptake of testing for type 2 diabetes or impaired glucose tolerance. Cochrane Database Syst Rev 2014;(3):CD009578.
- Carson MP, Frank MI, Keely E. Original research: Postpartum testing rates among women with a history of gestational diabetes–systematic review. Prim Care Diabetes 2013;7:177–86.
- Lawrence JM, Black MH, Hsu JW, et al. Prevalence and timing of postpartum glucose testing and sustained glucose dysregulation after gestational diabetes mellitus. Diabetes Care 2010;33:569–76.
- Halperin IJ, Sehgal P, Lowe J, et al. Increasing timely postpartum oral glucose tolerance test completion in women with gestational diabetes: A qualityimprovement initiative. Can J Diabetes 2015;39:451–6.
- Holt RI, Goddard JR, Clarke P, et al. A postnatal fasting plasma glucose is useful in determining which women with gestational diabetes should undergo a postnatal oral glucose tolerance test. Diabet Med 2003;20:594–8.
- Reinblatt SL, Morin L, Meltzer SJ. The importance of a postpartum 75 g oral glucose tolerance test in women with gestational diabetes. J Obstet Gynaecol Can 2006;28:690–4.
- Ferrara A, Peng T, Kim C. Trends in postpartum diabetes screening and subsequent diabetes and impaired fasting glucose among women with histories of gestational diabetes mellitus: A report from the Translating Research Into Action for Diabetes (TRIAD) Study. Diabetes Care 2009;32:269–74.
- McClean S, Farrar D, Kelly CA, et al. The importance of postpartum glucose tolerance testing after pregnancies complicated by gestational diabetes. Diabet Med 2010;27:650–4.
- Werner EF, Has P, Tarabulsi G, et al. Early postpartum glucose testing in women with gestational diabetes mellitus. Am J Perinatol 2016;33:966–71.
- Kim KS, Kim SK, Cho YW, et al. Diagnostic value of haemoglobin A1c in postpartum screening of women with gestational diabetes mellitus. Diabet Med 2016;33:1668–72.
- Su X, Zhang Z, Qu X, et al. Hemoglobin A1c for diagnosis of postpartum abnormal glucose tolerance among women with gestational diabetes mellitus: Diagnostic meta-analysis. PLoS ONE 2014;9:e102144.
- Claesson R, Ekelund M, Ignell C, et al. Role of HbA1c in post-partum screening of women with gestational diabetes mellitus. J Clin Transl Endocrinol 2015;2:21–5.
- Nohira T, Kim S, Nakai H, et al. Recurrence of gestational diabetes mellitus: Rates and risk factors from initial GDM and one abnormal GTT value. Diabetes Res Clin Pract 2006;71:75–81.
- Kim C, Berger DK, Chamany S. Recurrence of gestational diabetes mellitus: A systematic review. Diabetes Care 2007;30:1314–19.
- Kousta E, Efstathiadou Z, Lawrence NJ, et al. The impact of ethnicity on glucose regulation and the metabolic syndrome following gestational diabetes. Diabetologia 2006;49:36–40.
- Bo S, Monge L, Macchetta C, et al. Prior gestational hyperglycemia: A longterm predictor of the metabolic syndrome. J Endocrinol Invest 2004;27:629–35.
- Lauenborg J, Mathiesen E, Hansen T, et al. The prevalence of the metabolic syndrome in a danish population of women with previous gestational diabetes mellitus is three-fold higher than in the general population. J Clin Endocrinol Metab 2005;90:4004–10.
- Noctor E, Crowe C, Carmody LA, et al. ATLANTIC-DIP: Prevalence of metabolic syndrome and insulin resistance in women with previous gestational diabetes mellitus by International Association of Diabetes in Pregnancy Study Groups criteria. Acta Diabetol 2015;52:153–60.
- Fadl H, Magnuson A, Ostlund I, et al. Gestational diabetes mellitus and later cardiovascular disease: A Swedish population based case-control study. BJOG 2014;121:1530–6.
- Rivas AM, González N, González J. High frequency of diabetes in early postpartum assessment of women with gestational diabetes mellitus. Diabetes Metab Syndr 2007;1:159–65. https://www.infona.pl/resource/bwmeta1.element.elsevier-376ab781-3115-3818-8cf1-7c45db7405ed.
- Carr DB, Utzschneider KM, Hull RL, et al. Gestational diabetes mellitus increases the risk of cardiovascular disease in women with a family history of type 2 diabetes. Diabetes Care 2006;29:2078–83.
- Bao W, Tobias DK, Bowers K, et al. Physical activity and sedentary behaviors associated with risk of progression from gestational diabetes mellitus to type 2 diabetes mellitus: A prospective cohort study. JAMA Intern Med 2014;174:1047–55.
- Morton S, Kirkwood S, Thangaratinam S. Interventions to modify the progression to type 2 diabetes mellitus in women with gestational diabetes: A systematic review of literature. Curr Opin Obstet Gynecol 2014;26:476–86.
- Graco M, Garrard J, Jasper AE. Participation in physical activity: Perceptions of women with a previous history of gestational diabetes mellitus. Health Promot J Austr 2009;20:20–5.
- Symons Downs D, Ulbrecht JS. Understanding exercise beliefs and behaviors in women with gestational diabetes mellitus. Diabetes Care 2006;29:236–40.
- O’Reilly SL, Dunbar JA, Versace V, et al. Mothers after Gestational Diabetes in Australia (MAGDA): A randomised controlled trial of a Postnatal Diabetes Prevention program. PLoS Med 2016;13:e1002092.
- Perez-Ferre N, Del Valle L, Torrejon MJ, et al. Diabetes mellitus and abnormal glucose tolerance development after gestational diabetes: A three-year, prospective, randomized, clinical-based, Mediterranean lifestyle interventional study with parallel groups. Clin Nutr 2015;34:579–85.
- Boney CM, Verma A, Tucker R, et al. Metabolic syndrome in childhood: Association with birth weight, maternal obesity, and gestational diabetes mellitus. Pediatrics 2005;115:e290–6.
- Kim SY, England JL, Sharma JA, et al. Gestational diabetes mellitus and risk of childhood overweight and obesity in offspring: A systematic review. Exp Diabetes Res 2011;2011:541308.
- Philipps LH, Santhakumaran S, Gale C, et al. The diabetic pregnancy and offspring BMI in childhood: A systematic review and meta-analysis. Diabetologia 2011;54:1957–66.
- Burguet A. Long-term outcome in children of mothers with gestational diabetes. Diabetes Metab 2010;36:682–94.
- Thaware PK, McKenna S, Patterson CC, et al. Untreated mild hyperglycemia during pregnancy and anthropometric measures of obesity in offspring at age 5–7 years. Diabetes Care 2015;38:1701–6.
- Zhu Y, Olsen SF, Mendola P, et al. Growth and obesity through the first 7 y of life in association with levels of maternal glycemia during pregnancy: A prospective cohort study. Am J Clin Nutr 2016;103:794–800.
- Gillman MW, Oakey H, Baghurst PA, et al. Effect of treatment of gestational diabetes mellitus on obesity in the next generation. Diabetes Care 2010;33:964–8.
- Landon MB, Rice MM, Varner MW, et al. Mild gestational diabetes mellitus and long-term child health. Diabetes Care 2015;38:445–52.
- Malcolm JC, Lawson ML, Gaboury I, et al. Glucose tolerance of offspring of mother with gestational diabetes mellitus in a low-risk population. Diabet Med 2006;23:565–70.
- Silverman BL, Rizzo TA, Cho NH, et al. Long-term effects of the intrauterine environment. The Northwestern University Diabetes in Pregnancy Center. Diabetes Care 1998;21:B142–9.
- Knorr S, Clausen TD, Vlachova Z, et al. Academic achievement in primary school in offspring born to mothers with type 1 diabetes (the EPICOM Study): A registerbased prospective cohort study. Diabetes Care 2015;38:1238–44.
- Xiang AH, Wang X, Martinez MP, et al. Association of maternal diabetes with autism in offspring. JAMA 2015;313:1425–34.
- Krakowiak P,Walker CK, Bremer AA, et al. Maternal metabolic conditions and risk for autism and other neurodevelopmental disorders. Pediatrics 2012;129:e1121–8.
- Damm P, Mathiesen ER, Petersen KR, et al. Contraception after gestational diabetes. Diabetes Care 2007;30(Suppl. 2):S236–41.
- Kjos SL, Peters RK, Xiang A, et al. Contraception and the risk of type 2 diabetes mellitus in Latina women with prior gestational diabetes mellitus. JAMA 1998;280:533–8.
- MacNeill S, Dodds L, Hamilton DC, et al. Rates and risk factors for recurrence of gestational diabetes. Diabetes Care 2001;24:659–62.
- Gaudier FL, Hauth JC, Poist M, et al. Recurrence of gestational diabetes mellitus. Obstet Gynecol 1992;80:755–8.
- McElvy SS, Miodovnik M, Rosenn B, et al. A focused preconceptional and early pregnancy program in women with type 1 diabetes reduces perinatal mortality and malformation rates to general population levels. J Matern Fetal Med 2000;9:14–20.
- Suhonen L, Hiilesmaa V, Teramo K. Glycaemic control during early pregnancy and fetal malformations in women with type I diabetes mellitus. Diabetologia 2000;43:79–82.
- Guerin A, Nisenbaum R, Ray JG. Use of maternal GHb concentration to estimate the risk of congenital anomalies in the offspring of women with prepregnancy diabetes. Diabetes Care 2007;30:1920–5.
- Hiilesmaa V, Suhonen L, Teramo K. Glycaemic control is associated with preeclampsia but not with pregnancy-induced hypertension in women with type I diabetes mellitus. Diabetologia 2000;43:1534–9.
- Hsu CD, Tan HY, Hong SF, et al. Strategies for reducing the frequency of preeclampsia in pregnancies with insulin-dependent diabetes mellitus. Am J Perinatol 1996;13:265–8.
- Edison RJ, MuenkeM. Central nervous system and limb anomalies in case reports of first-trimester statin exposure. N Engl J Med 2004;350:1579–82.
- Hod M, Mathiesen ER, Jovanovicˇ L, et al. A randomized trial comparing perinatal outcomes using insulin detemir or neutral protamine Hagedorn in type 1 diabetes. J Matern Fetal Neonatal Med 2014;27:7–13.
- Griffin ME, Coffey M, Johnson H, et al. Universal vs. risk factor-based screening for gestational diabetes mellitus: Detection rates, gestation at diagnosis and outcome. Diabet Med 2000;17:26–32.
- Horta BL, Loret de Mola C, Victora CG. Long-term consequences of breastfeeding on cholesterol, obesity, systolic blood pressure and type 2 diabetes: A systematic review and meta-analysis. Acta Paediatr 2015;104:30–7.
- Aroda VR, Christophi CA, Edelstein SL, et al. The effect of lifestyle intervention and metformin on preventing or delaying diabetes among women with and without gestational diabetes: The Diabetes Prevention Program outcomes study 10-year follow-up. J Clin Endocrinol Metab 2015;100:1646–53.
- Ratner RE, Christophi CA, Metzger BE, et al. Prevention of diabetes in women with a history of gestational diabetes: Effects of metformin and lifestyle interventions. J Clin Endocrinol Metab 2008;93:4774–9.
- Metzger BE, Gabbe SG, Persson B, et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010;33:676–82.
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med 2009;6:e1000097.